13.3
Impact Factor
Theranostics 2021; 11(7):3452-3471. doi:10.7150/thno.53572 This issue Cite
Research Paper
Mutation of SPINOPHILIN (PPP1R9B) found in human tumors promotes the tumorigenic and stemness properties of cells
1. Instituto de Biomedicina de Sevilla, IBIS, Hospital Universitario Virgen del Rocio, Consejo Superior de Investigaciones Científicas, Universidad de Sevilla, Avda. Manuel Siurot s/n, 41013 Seville, Spain.
2. CIBERONC, Instituto de Salud Carlos III, 28029 Madrid, Spain.
3. Centro Andaluz de Biología del Desarrollo (CABD), CSIC-Universidad Pablo de Olavide, Sevilla, Spain.
Abstract
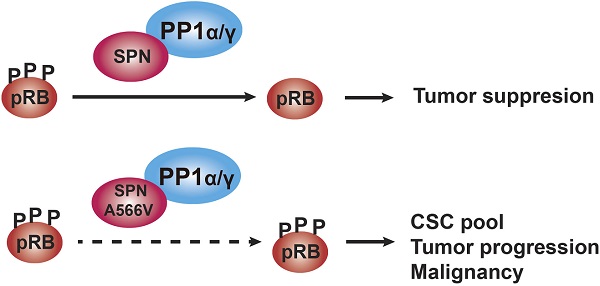
Rationale: SPINOPHILIN (SPN, PPP1R9B) is an important tumor suppressor involved in the progression and malignancy of different tumors depending on its association with protein phosphatase 1 (PP1) and the ability of the PP1-SPN holoenzyme to dephosphorylate retinoblastoma (pRB).
Methods: We performed a mutational analysis of SPN in human tumors, focusing on the region of interaction with PP1 and pRB. We explored the effect of the SPN-A566V mutation in an immortalized non-tumorigenic cell line of epithelial breast tissue, MCF10A, and in two different p53-mutated breast cancer cells lines, T47D and MDA-MB-468.
Results: We characterized an oncogenic mutation of SPN found in human tumor samples, SPN-A566V, that affects both the SPN-PP1 interaction and its phosphatase activity. The SPN-A566V mutation does not affect the interaction of the PP1-SPN holoenzyme with pocket proteins pRB, p107 and p130, but it affects its ability to dephosphorylate them during G0/G1 and G1, indicating that the PP1-SPN holoenzyme regulates cell cycle progression. SPN-A566V also promoted stemness, establishing a connection between the cell cycle and stem cell biology via pocket proteins and PP1-SPN regulation. However, only cells with both SPN-A566V and mutant p53 have increased tumorigenic and stemness properties.
Conclusions: SPN-A566V, or other equivalent mutations, could be late events that promote tumor progression by increasing the CSC pool and, eventually, the malignant behavior of the tumor.
Keywords: SPINOPHILIN, PP1, cancer stem cell, tumorigenesis, stem cell phenotype, pRB, pocket proteins
 Global reach, higher impact
Global reach, higher impact