Impact Factor
Theranostics 2021; 11(8):3552-3564. doi:10.7150/thno.53225 This issue Cite
Research Paper
Vitamin C sensitizes triple negative breast cancer to PI3K inhibition therapy
1. John P. Hussman Institute for Human Genomics, Dr. John T. Macdonald Foundation Department of Human Genetics, University of Miami Miller School of Medicine, Miami, Florida.
2. Sylvester Comprehensive Cancer Center, University of Miami Miller School of Medicine, Miami, Florida.
3. Braman Family Breast Cancer Institute at Sylvester, University of Miami Miller School of Medicine, Miami, Florida.
Abstract
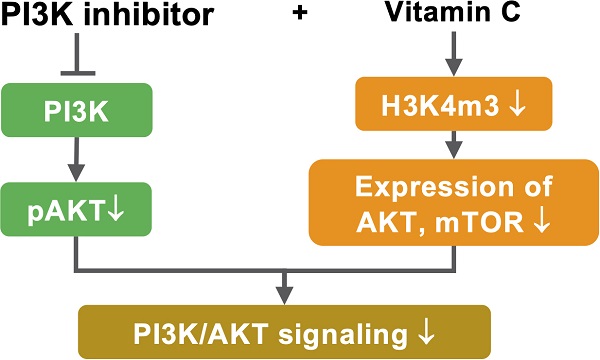
Rationale: The clinical use of PI3K inhibitors, such as buparlisib, has been plagued with toxicity at effective doses. The aim of this study is to determine if vitamin C, a potent epigenetic regulator, can improve the therapeutic outcome and reduce the dose of buparlisib in treating PIK3CA-mutated triple negative breast cancer (TNBC).
Methods: The response of TNBC cells to buparlisib was assessed by EC50 measurements, apoptosis assay, clonogenic assay, and xenograft assay in mice. Molecular approaches including Western blot, immunofluorescence, RNA sequencing, and gene silencing were utilized as experimental tools.
Results: Treatment with buparlisib at lower doses, along with vitamin C, induced apoptosis and inhibited the growth of TNBC cells in vitro. Vitamin C via oral delivery rendered a sub-therapeutic dose of buparlisib able to inhibit TNBC xenograft growth and to markedly block metastasis in mice. We discovered that buparlisib and vitamin C coordinately reduced histone H3K4 methylation by enhancing the nuclear translocation of demethylase, KDM5, and by serving as a cofactor to promote KDM5-mediated H3K4 demethylation. The expression of genes in the PI3K pathway, such as AKT2 and mTOR, was suppressed by vitamin C in a KDM5-dependent manner. Vitamin C and buparlisib cooperatively blocked AKT phosphorylation. Inhibition of KDM5 largely abolished the effect of vitamin C on the response of TNBC cells to buparlisib. Additionally, vitamin C and buparlisib co-treatment changed the expression of genes, including PCNA and FILIP1L, which are critical to cancer growth and metastasis.
Conclusion: Vitamin C can be used to reduce the dosage of buparlisib needed to produce a therapeutic effect, which could potentially ease the dose-dependent side effects in patients.
 Global reach, higher impact
Global reach, higher impact