13.3
Impact Factor
Theranostics 2021; 11(8):3642-3660. doi:10.7150/thno.53089 This issue Cite
Research Paper
Nanodefensin-encased hydrogel with dual bactericidal and pro-regenerative functions for advanced wound therapy
1. Department of Anesthesiology and Intensive Care, The First Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou 310003, China.
2. Department of Polymer Science and Engineering, Zhejiang University, Hangzhou 310027, China.
3. Laboratory of Computational and Quantitative Biology (LCQB), Institute of Biology, Paris-Seine, Sorbonne University, Paris, 75006, France.
4. Key Laboratory of Medical Molecular Virology (MOE/NHC/CAMS), School of Basic Medical Sciences, Fudan University, Shanghai, 200032, China.
#These authors contributed equally to this work.
Abstract
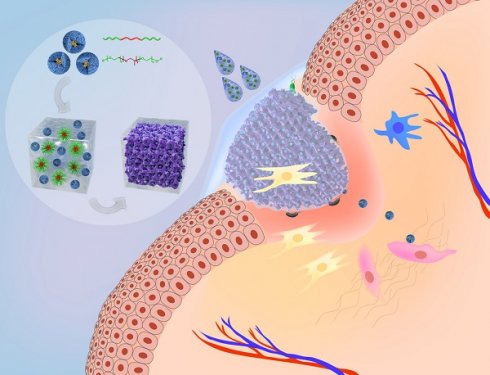
Background: Host defense peptides (HDPs) have emerged as a novel therapeutic paradigm for wound management; however, their clinical applications remain a challenge owing to their poor pharmacological properties and lack of suitable pharmaceutical formulations. Nanodefensin (ND), a nanoengineered human α-defensin 5 (HD5), has shown improved pharmacological properties relative to the parent compound. In this study, we engineered a nanodefensin-encased hydrogel (NDEFgel), investigated the effects of NDEFgel on wound healing, and elucidated underlying mechanisms.
Method: ND was chemically synthesized and tested functions by in vitro antimicrobial and scratch assays and western blotting. Different NDEFgels were evaluated by in vitro characterizations including degradation, drug release and antimicrobial activity. In full-thickness excisional murine models, the optimal NDEFgel was directly applied onto wound sites, and the efficacy was assessed. Moreover, the underlying mechanisms of pro-regenerative effect developed by NDEFgel were also explored.
Results: Apart from bactericidal effects, ND modulated fibroblast behaviors by promoting migration and differentiation. Among the tested hydrogels, the Pluronic F127 (Plu) hydrogel represented the most desirable carrier for ND delivery owing to its favorable controlled release and compatibility with ND. Local treatment of NDEFgel on the wound bed resulted in accelerated wound regeneration and attenuated bacterial burden. We further demonstrated that NDEFgel therapy significantly upregulated genes related to collagen deposition and fibroblasts, and increased the expression of myofibroblasts and Rac1. We therefore found that Rac1 is a critical factor in the ND-induced modulation of fibroblast behaviors in vitro through a Rac1-dependent cytoskeletal rearrangement.
Conclusion: Our results indicate that NDEFgel may be a promising dual-action therapeutic option for advanced wound management in the future.
Keywords: host defense peptides, wound healing, regenerative medicine, biomaterials, pharmaceutical formulation
 Global reach, higher impact
Global reach, higher impact