13.3
Impact Factor
Theranostics 2021; 11(8):3725-3741. doi:10.7150/thno.53644 This issue Cite
Research Paper
Dual-ligand supramolecular nanofibers inspired by the renin-angiotensin system for the targeting and synergistic therapy of myocardial infarction
1. Guangdong Provincial Biomedical Engineering Technology Research Center for Cardiovascular Disease, Department of Cardiology and Laboratory of Heart Center, Sino-Japanese Cooperation Platform for Translational Research in Heart Failure, Zhujiang Hospital, Southern Medical University, Guangzhou 510280, China.
2. Shunde Hospital, Southern Medical University, the First People's Hospital of Shunde, Foshan 528300, China.
3. School of Pharmaceutical Sciences, Southern Medical University, Guangzhou 510515, China.
4. Department of Cardiology, Nanfang Hospital, Southern Medical University; Guangzhou 510515, China.
5. Key Laboratory of Bioactive Materials, Ministry of Education, State Key Laboratory of Medicinal Chemical Biology, College of Life Sciences, Nankai University, Tianjin 300071, China.
6. Jiangsu Center for the Collaboration and Innovation of Cancer Biotherapy, Cancer Institute, Xuzhou Medical University, Xuzhou 221004, Jiangsu, China.
#These authors contributed equally to this work.
Abstract
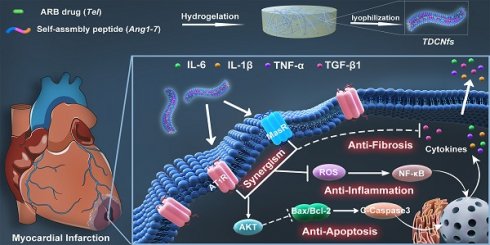
Rationale: The compensatory activation of the renin-angiotensin system (RAS) after myocardial infarction (MI) plays a crucial role in the pathogenesis of heart failure. Most existing studies on this subject focus on mono- or dual-therapy of blocking the RAS, which exhibit limited efficacy and often causes serious adverse reactions. Few studies have been conducted on targeted therapy based on the activated RAS post-MI. Thus, the development of multiple-functional nanomedicine with concurrent targeting ability and synergistic therapeutic effect against RAS may show great promise in improving cardiac function post-MI.
Methods: We utilized a cooperative self-assembly strategy constructing supramolecular nanofibers— telmisartan-doped co-assembly nanofibers (TDCNfs) to counter-regulate RAS through targeted delivery and combined therapy. TDCNfs were prepared through serial steps of solvent exchange, heating incubation, gelation, centrifugation, and lyophilization, in which the telmisartan was doped in the self-assembly process of Ang1-7 to obtain the co-assembly nanofibers wherein they act as both therapeutic agents and target-guide agents.
Results: TDCNfs exhibited the desired binding affinity to the two different receptors, AT1R and MasR. Through the dual ligand-receptor interactions to mediate the coincident downstream pathways, TDCNfs not only displayed favorably targeted properties to hypoxic cardiomyocytes, but also exerted synergistic therapeutic effects in apoptosis reduction, inflammatory response alleviation, and fibrosis inhibition in vitro and in vivo, significantly protecting cardiac function and mitigating post-MI adverse outcomes.
Conclusion: A dual-ligand nanoplatform was successfully developed to achieve targeted and synergistic therapy against cardiac deterioration post-MI. We envision that the integration of multiple therapeutic agents through supramolecular self-assembly would offer new insight for the systematic and targeted treatment of cardiovascular diseases.
Keywords: supramolecular self-assembly, myocardial infarction, renin-angiotensin system (RAS), targeted therapy, synergistic effect
 Global reach, higher impact
Global reach, higher impact