13.3
Impact Factor
Theranostics 2021; 11(8):3830-3838. doi:10.7150/thno.49837 This issue Cite
Research Paper
Sunitinib-induced cardiac hypertrophy and the endothelin axis
1. Université de Paris, PARCC, INSERM, F-75015 Paris.
2. Aix-Marseille Université, CNRS, CRMBM, UMR 7339, Marseille, France.
3. INSERM U1016, Institut Cochin, Université de Paris, Paris 75014, France.
4. Équipe labellisée par la Ligue contre le Cancer.
5. Équipe labellisée par la Fondation pour la Recherche Médicale.
6. University/British Heart Foundation Centre of Research Excellence, The Queen's Medical Research Institute, University of Edinburgh, United Kingdom.
7. Service de radiologie, AP-HP, Hôpital européen Georges Pompidou, F-75015 Paris.
* These authors contributed equally to this work
Abstract
Anti-angiogenics drugs in clinical use for cancer treatment induce cardiotoxic side effects. The endothelin axis is involved in hypertension and cardiac remodelling, and addition of an endothelin receptor antagonist to the anti-angiogenic sunitinib was shown to reduce cardiotoxicity of sunitinib in mice. Here, we explored further the antidote effect of the endothelin receptor antagonist macitentan in sunitinib-treated animals on cardiac remodeling.
Methods: Tumor-bearing mice treated per os daily by sunitinib or vehicle were imaged before and after 1, 3 and 6 weeks of treatment by positron emission tomography using [18F]fluorodeoxyglucose and by echocardiography. Non-tumor-bearing animals were randomly assigned to be treated per os daily by vehicle or sunitinib or macitentan or sunitinib+macitentan, and imaged by echocardiography after 5 weeks. Hearts were harvested for histology and molecular analysis at the end of in vivo exploration.
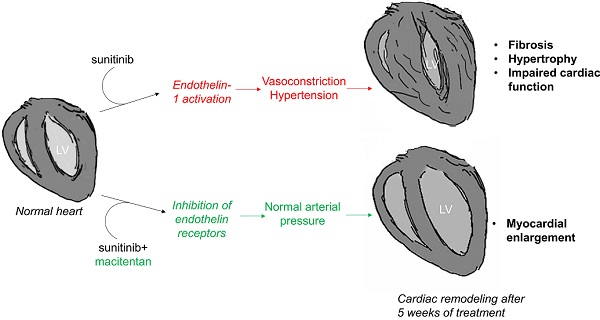
Results: Sunitinib treatment increases left ventricular mass and ejection fraction and induces cardiac fibrosis. Sunitinib also induces an early increase in cardiac uptake of [18F]fluorodeoxyglucose, which is significantly correlated with increased left ventricular mass at the end of treatment. Co-administration of macitentan prevents sunitinib-induced hypertension, increase in ejection fraction and cardiac fibrosis, but fails to prevent increase of the left ventricular mass.
Conclusion: Early metabolic changes predict sunitinib-induced cardiac remodeling. Endothelin blockade can prevent some but not all cardiotoxic side-effects of sunitinib, in particular left ventricle hypertrophy that appears to be induced by sunitinib through an endothelin-independent mechanism.
Keywords: cardiotoxicity, sunitinib, endothelin, cardiac hypertrophy, metabolic imaging.
 Global reach, higher impact
Global reach, higher impact