Impact Factor
Theranostics 2021; 11(8):3868-3881. doi:10.7150/thno.53347 This issue Cite
Research Paper
KDM6B-mediated histone demethylation of LDHA promotes lung metastasis of osteosarcoma
1. Department of Orthopedics, The Eighth Affiliated Hospital of Sun Yat-sen University, Shenzhen 518003, China.
2. Institute of Interdisciplinary Integrative Medicine Research, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China.
3. Department of Orthopaedics, The Second Affiliated Hospital of Guangzhou Medical University, 250 Changgang Eastern Road, Guangzhou 510260, China.
4. Department of Musculoskeletal Surgery, Fudan University Shanghai Cancer center, Shanghai Medical College, Fudan University, Shanghai 200032, China.
5. Department of Plastic and Reconstructive Surgery, Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, China.
* These authors contributed equally to this work.
# These authors contributed equally to this work.
Abstract
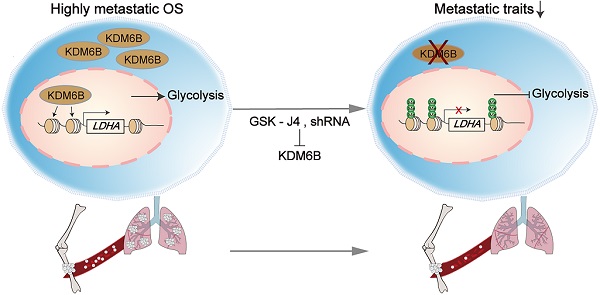
Rationale: Osteosarcoma (OS), the most common type of bone tumor, which seriously affects the patients' limb function and life quality. OS has a strong tendency of lung metastasis, and the five-year survival rate of patients with metastatic osteosarcoma is less than 20%. Thus, new treatment targets and strategies are urgently needed.
Methods: The expression of the histone demethylase KDM6B and H3K27me3 levels in OS specimens were analyzed using quantitative PCR and immunohistochemical assays. The biological functions of KDM6B were determined using in vitro transwell, wound healing assays, and an in vivo orthotopic injection-induced lung metastasis model. Subsequently, chromatin immunoprecipitation sequencing (ChIP-seq) combined with transcriptomic RNA sequencing (RNA-seq), and subsequent ChIP-qPCR, western blot, and aerobic glycolysis assays were used to explore the mechanism of KDM6B function and validate the candidate target gene of KDM6B.
Results: KDM6B expression was significantly upregulated in OS patients, and high KDM6B expression was associated with poorer prognosis in OS patients. Targeting KDM6B significantly inhibited OS cell migration in vitro and lung metastasis in vivo. RNA-seq and ChIP-seq analysis revealed that KDM6B increases lactate dehydrogenase LDHA expression in OS cells by directly mediating H3K27me3 demethylation. The phenotypes of inhibited cell metastasis in KDM6B-knockdown OS cells was reversed upon overexpression of LDHA. Finally, a small molecule inhibitor targeting KDM6B significantly inhibited OS cell migration in vitro and lung metastasis in vivo.
Conclusions: Collectively, we elucidated that upregulated KDM6B facilitates tumor metastasis in OS via modulating LDHA expression. Our findings deepen the recognition of OS metastasis mechanism and suggest that KDM6B might be a new potential therapeutic target for the treatment of OS (especially highly metastatic OS).
Keywords: Osteosarcoma, Lung metastasis, H3K27me3 demethylation, KDM6B, LDHA.
 Global reach, higher impact
Global reach, higher impact