13.3
Impact Factor
Theranostics 2021; 11(8):3932-3947. doi:10.7150/thno.53412 This issue Cite
Research Paper
Exosomal miR-500a-5p derived from cancer-associated fibroblasts promotes breast cancer cell proliferation and metastasis through targeting USP28
1. Department of Breast Surgery, General Surgery, Qilu Hospital of Shandong University.
2. Pathology Tissue Bank, Qilu Hospital of Shandong University.
3. Research Institute of Breast Cancer, Shandong University.
*These authors contributed equally to this work.
Abstract
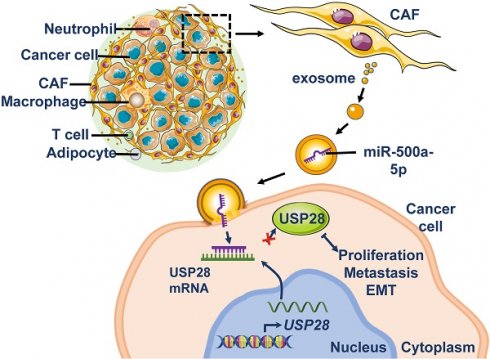
The tumor microenvironment contributes to tumor progression and metastasis. Cancer-associated fibroblasts (CAFs) form a major cellular component of the tumor microenvironment. In this study, we further explored the mechanisms underlying the tumor-promoting roles of CAFs.
Methods: Patient-derived CAFs and normal fibroblasts (NFs) were isolated from breast carcinomas and adjacent normal breast tissue. Exosomes were isolated by ultracentrifugation and CAF-derived exosomal microRNAs were screened using next-generation sequencing technology. MiR-500a-5p expression was assessed by quantitative real-time polymerase chain reaction (qRT-PCR) and in situ hybridization; Tumor cell proliferation was determined by MTT assays and three-dimensioned (3D) cultures, and tumor metastasis was determined by Transwell assays in vitro. In vivo assays were performed in a nude mouse subcutaneous xenograft model.
Results: We confirmed that CAF-derived exosomes significantly promoted the proliferation and metastasis of breast cancer cells. MiR-500a-5p was highly expressed in MDA-MB-231 and MCF7 cells treated with CAF-derived exosomes. The upregulation of miR-500a-5p was also confirmed in CAFs and CAF-derived exosomes. MiR-500a-5p was transferred from CAFs to the cancer cells, and subsequently promoted proliferation and metastasis by binding to ubiquitin-specific peptidase 28 (USP28).
Conclusions: The present study demonstrates that CAFs promote breast cancer progression and metastasis via exosomal miR-500a-5p and indicate that inhibiting CAF-derived miR-500a-5p is an alternative modality for the treatment of breast cancer.
Keywords: cancer-associated fibroblasts, exosome, miR-500a-5p, breast cancer, USP28
 Global reach, higher impact
Global reach, higher impact