Impact Factor
Theranostics 2021; 11(8):3948-3960. doi:10.7150/thno.54959 This issue Cite
Research Paper
Injectable conductive hydrogel can reduce pacing threshold and enhance efficacy of cardiac pacemaker
1. Toronto General Hospital Research Institute, Division of Cardiovascular Surgery, University Health Network, Toronto, Ontario, Canada.
2. Department of Cardiovascular Surgery, Changhai Hospital, Naval Medical University, Shanghai, China.
3. Department of Chemical Engineering, National Tsing Hua University, Hsinchu, Taiwan (ROC).
Abstract
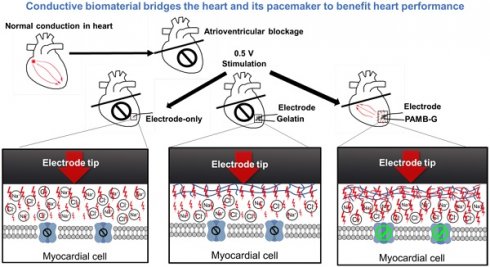
Background: Pacemaker implantation is currently used in patients with symptomatic bradycardia. Since a pacemaker is a lifetime therapeutic device, its energy consumption contributes to battery exhaustion, along with its voltage stimulation resulting in local fibrosis and greater resistance, which are all detrimental to patients. The possible resolution for those clinical issues is an injection of a conductive hydrogel, poly-3-amino-4-methoxybenzoic acid-gelatin (PAMB-G), to reduce the myocardial threshold voltage for pacemaker stimulation. Methods: PAMB-G is synthesized by covalently linking PAMB to gelatin, and its conductivity is measured using two-point resistivity. Rat hearts are injected with gelatin or PAMB-G, and pacing threshold is evaluated using electrocardiogram and cardiac optical mapping. Results: PAMB-G conductivity is 13 times greater than in gelatin. The ex vivo model shows that PAMB-G significantly enhances cardiac tissue stimulation. Injection of PAMB-G into the stimulating electrode location at the myocardium has a 4 times greater reduction of pacing threshold voltage, compared with electrode-only or gelatin-injected tissues. Multi-electrode array mapping reveals that the cardiac conduction velocity of PAMB-G group is significantly faster than the non- or gelatin-injection groups. PAMB-G also reduces pacing threshold voltage in an adenosine-induced atrial-ventricular block rat model. Conclusion: PAMB-G hydrogel reduces cardiac pacing threshold voltage, which is able to enhance pacemaker efficacy.
Keywords: conductive biomaterials, pacing threshold, tissue resistance, pacemaker, cardiac arrhythmia.
 Global reach, higher impact
Global reach, higher impact