13.3
Impact Factor
Theranostics 2021; 11(10):4688-4698. doi:10.7150/thno.55856 This issue Cite
Research Paper
Synthesis of RNA-based gene regulatory devices for redirecting cellular signaling events mediated by p53
1. Guangdong and Shenzhen Key Laboratory of Male Reproductive Medicine and Genetics, Institute of Urology, Peking University Shenzhen Hospital, Shenzhen-Peking University-the Hong Kong University of Science and Technology Medical Center, Shenzhen 518000, China.
2. Institute of Translational Medicine, Shenzhen Second People's Hospital, The First Affiliated Hospital of Shenzhen University, Shenzhen 518039, China.
Abstract
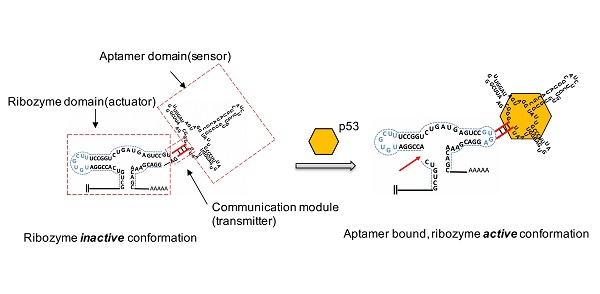
Rationale: The p53 gene is a well-known tumor suppressor, and its mutation often contributes to the occurrence and development of tumors. Due to the diversity and complexity of p53 mutations, there is still no effective p53 gene therapy. In this study, we designed and constructed an aptazyme switch that could effectively sense cellular wild-type p53 protein and regulate downstream gene function flexibly. The application of this artificial device in combination with Cre-LoxP and dCas9-VP64 tools achieved a precisely targeted killing effect on tumor cells.
Methods: The affinity of the aptamer to p53 protein was verified by SPR. p53 aptazyme and gene circuits were chemically synthesized. The function of the gene circuit was detected by cell proliferation assay, apoptosis assay and Western blot. The nude mouse transplantation tumor experiment was used to evaluate the inhibitory effect of gene circuits on tumor cells in vivo.
Results: The results of the SPR experiment showed that the p53 aptamer RNA sequence had a robust binding effect with p53 protein. The p53 aptazyme could efficiently sense wild-type p53 protein and initiate self-cleavage in cells. The Cre-p53 aptazyme gene circuit and dCas9-VP64/sgRNA mediated gene circuit designed based on p53 aptazyme significantly inhibited the growth and promoted the apoptosis of wild-type p53-deficient cancer cells in vitro. In addition, the gene circuits also had a significant inhibitory effect on tumors in vivo.
Conclusion: The study developed a novel and efficient ribozyme switch for p53-specific recognition and provided a modular strategy for aptazyme binding to cellular proteins. In addition, the p53 aptazyme successfully inhibited tumor growth through a combined application with other synthetic biological tools, providing a new perspective for cancer therapy.
Keywords: synthetic biology, gene circuit, p53 aptazyme, Cre/loxP, CRISPR.
 Global reach, higher impact
Global reach, higher impact