Impact Factor
Theranostics 2021; 11(10):4728-4742. doi:10.7150/thno.56598 This issue Cite
Research Paper
GPR43 deficiency protects against podocyte insulin resistance in diabetic nephropathy through the restoration of AMPKα activity
1. Institute of Nephrology, Zhongda Hospital, School of Medicine, Southeast University, Nanjing, 210009, China.
2. John Moorhead Research Laboratory, Department of Renal Medicine, University College London (UCL) Medical School, Royal Free Campus, London, NW3 2PF, UK.
Abstract
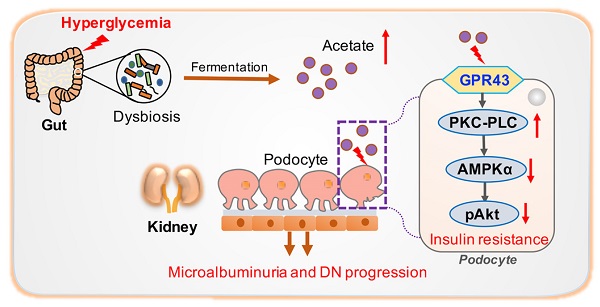
Rationale: Albuminuria is an early clinical feature in the progression of diabetic nephropathy (DN). Podocyte insulin resistance is a main cause of podocyte injury, playing crucial roles by contributing to albuminuria in early DN. G protein-coupled receptor 43 (GPR43) is a metabolite sensor modulating the cell signalling pathways to maintain metabolic homeostasis. However, the roles of GPR43 in podocyte insulin resistance and its potential mechanisms in the development of DN are unclear.
Methods: The experiments were conducted by using kidney tissues from biopsied DN patients, streptozotocin (STZ) induced diabetic mice with or without global GPR43 gene knockout, diabetic rats treated with broad-spectrum oral antibiotics or fecal microbiota transplantation, and cell culture model of podocytes. Renal pathological injuries were evaluated by periodic acid-schiff staining and transmission electron microscopy. The expression of GPR43 with other podocyte insulin resistance related molecules was checked by immunofluorescent staining, real-time PCR, and Western blotting. Serum acetate level was examined by gas chromatographic analysis. The distribution of gut microbiota was measured by 16S ribosomal DNA sequencing with faeces.
Results: Our results demonstrated that GPR43 expression was increased in kidney samples of DN patients, diabetic animal models, and high glucose-stimulated podocytes. Interestingly, deletion of GPR43 alleviated albuminuria and renal injury in diabetic mice. Pharmacological inhibition and knockdown of GPR43 expression in podocytes increased insulin-induced Akt phosphorylation through the restoration of adenosine 5'-monophosphate-activated protein kinase α (AMPKα) activity. This effect was associated with the suppression of AMPKα activity through post-transcriptional phosphorylation via the protein kinase C-phospholipase C (PKC-PLC) pathway. Antibiotic treatment-mediated gut microbiota depletion, and faecal microbiota transplantation from the healthy donor controls substantially improved podocyte insulin sensitivity and attenuated glomerular injury in diabetic rats accompanied by the downregulation of the GPR43 expression and a decrease in the level of serum acetate.
Conclusion: These findings suggested that dysbiosis of gut microbiota-modulated GPR43 activation contributed to albuminuria in DN, which could be mediated by podocyte insulin resistance through the inhibition of AMPKα activity.
Keywords: diabetic nephropathy, podocyte insulin resistance, GPR43, AMPKα activity, gut microbiota dysbiosis
 Global reach, higher impact
Global reach, higher impact