13.3
Impact Factor
Theranostics 2021; 11(10):4809-4824. doi:10.7150/thno.55814 This issue Cite
Research Paper
WD repeat domain 5 promotes chemoresistance and Programmed Death-Ligand 1 expression in prostate cancer
1. Department of Urology, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China.
2. Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation, Sun Yat-Sen Memorial Hospital, Sun Yat-Sen University, Guangzhou 510120, China.
3. State Key Laboratory of Oncology in South China & Collaborative Innovation Center of Cancer Medicine, Sun Yat-sen University Cancer Center, Guangzhou, 510060, China.
4. Department of Medical Oncology, Sun Yat-sen University Cancer Center, Guangzhou, 510060, China.
5. Department of Urology, The Affiliated Kashi Hospital, Sun Yat-sen University, Kashi, China.
#These authors contributed equally to this paper.
Abstract
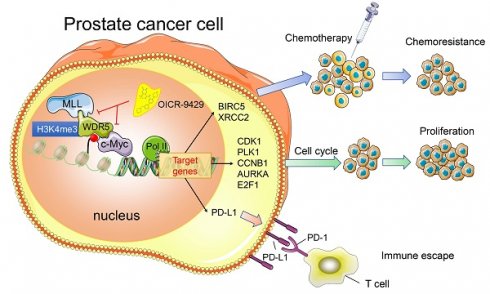
Purpose: Advanced prostate cancer (PCa) has limited treatment regimens and shows low response to chemotherapy and immunotherapy, leading to poor prognosis. Histone modification is a vital mechanism of gene expression and a promising therapy target. In this study, we characterized WD repeat domain 5 (WDR5), a regulator of histone modification, and explored its potential therapeutic value in PCa.
Experimental Design: We characterized specific regulators of histone modification, based on TCGA data. The expression and clinical features of WDR5 were analyzed in two dependent cohorts. The functional role of WDR5 was further investigated with siRNA and OICR-9429, a small molecular antagonist of WDR5, in vitro and in vivo. The mechanism of WDR5 was explored by RNA-sequencing and chromatin immunoprecipitation (ChIP).
Results: WDR5 was overexpressed in PCa and associated with advanced clinicopathological features, and predicted poor prognosis. Both inhibition of WDR5 by siRNA and OICR-9429 could reduce proliferation, and increase apoptosis and chemosensitivity to cisplatin in vitro and in vivo. Interestingly, targeting WDR5 by siRNA and OICR-9429 could block IFN-γ-induced PD-L1 expression in PCa cells. Mechanistically, we clarified that some cell cycle, anti-apoptosis, DNA repair and immune related genes, including AURKA, CCNB1, E2F1, PLK1, BIRC5, XRCC2 and PD-L1, were directly regulated by WDR5 and OICR-9429 in H3K4me3 and c-Myc dependent manner.
Conclusions: These data revealed that targeting WDR5 suppressed proliferation, enhanced apoptosis, chemosensitivity to cisplatin and immunotherapy in PCa. Therefore, our findings provide insight into OICR-9429 is a multi-potency and promising therapy drug, which improves the antitumor effect of cisplatin or immunotherapy in PCa.
Keywords: Prostate cancer, WDR5, chemoresistance, OICR-9429, PD-L1
 Global reach, higher impact
Global reach, higher impact