13.3
Impact Factor
Theranostics 2021; 11(11):5127-5142. doi:10.7150/thno.54741 This issue Cite
Research Paper
Kinomic profile in patient-derived glioma cells during hypoxia reveals c-MET-PI3K dependency for adaptation
1. School of Biological Sciences, Nanyang Technological University Singapore, Singapore 637551, Singapore.
2. Lee Kong Chian School of Medicine, Nanyang Technological University Singapore, Singapore 308232, Singapore.
3. Neuro-Oncology Research Laboratory, Department of Research, National Neuroscience Institute, Singapore 308433, Singapore.
4. Department of Neurosurgery, National Neuroscience Institute, Singapore 308433, Singapore.
5. Duke-National University of Singapore Medical School, Singapore 169857, Singapore.
6. Department of Physiology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore 117593, Singapore.
Abstract
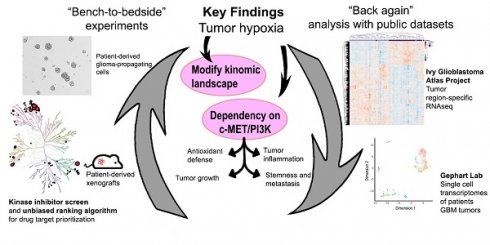
Hypoxic microenvironment is a hallmark of solid tumors, especially glioblastoma. The strong reliance of glioma-propagating cells (GPCs) on hypoxia-induced survival advantages is potentially exploitable for drug development.
Methods: To identify key signaling pathways for hypoxia adaptation by patient-derived GPCs, we performed a kinase inhibitor profiling by screening 188 small molecule inhibitors against 130 different kinases in normoxia and hypoxia. Potential kinase candidates were prioritized for in vitro and in vivo investigations using a ranking algorithm that integrated information from the kinome connectivity network and estimated patients' survival based on expression status.
Results: Hypoxic drug screen highlighted extensive modifications of kinomic landscape and a crucial functionality of c-MET-PI3K. c-MET inhibitors diminished phosphorylation of c-MET and PI3K in GPCs subjected to hypoxia, suggesting its role in the hypoxic adaptation of GPCs. Mechanistically, the inhibition of c-MET and PI3K impaired antioxidant defense, leading to oxidative catastrophe and apoptosis. Repurposed c-MET inhibitors PF04217903 and tivantinib exhibited hypoxic-dependent drug synergism with temozolomide, resulting in reduced tumor load and growth of GPC xenografts. Detailed analysis of bulk and single-cell glioblastoma transcriptomes associates the cellular subpopulation over-expressing c-MET with inflamed, hypoxic, metastatic, and stem-like phenotypes.
Conclusions: Thus, our “bench to bedside (the use of patient-derived GPCs and xenografts for basic research) and back (validation with independent glioblastoma transcriptome databases)” analysis unravels the novel therapeutic indications of c-MET and PI3K/Akt inhibitors for the treatment of glioblastoma, and potentially other cancers, in the hypoxic tumor microenvironment.
Keywords: glioblastoma, HIF-1α, oxidative stress, PI3K, tumor microenvironment
 Global reach, higher impact
Global reach, higher impact