13.3
Impact Factor
Theranostics 2021; 11(12):5713-5727. doi:10.7150/thno.55250 This issue Cite
Research Paper
Quantitative self-assembly of pure drug cocktails as injectable nanomedicines for synergistic drug delivery and cancer therapy
1. The First Affiliated Hospital, Zhejiang University School of Medicine; NHC Key Laboratory of Combined Multi-Organ Transplantation; Key Laboratory of Organ Transplantation, Research Center for Diagnosis and Treatment of Hepatobiliary Diseases, Zhejiang Province, Hangzhou, PR China.
2. Department of Medical Oncology, Sir Run Run Shaw Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, 310003, PR China.
3. Department of Chemical Engineering, Zhejiang University, Hangzhou, 310027, PR China.
4. Central Laboratory, Shanghai Pudong New Area People's Hospital, Shanghai, 201299, PR China.
Abstract
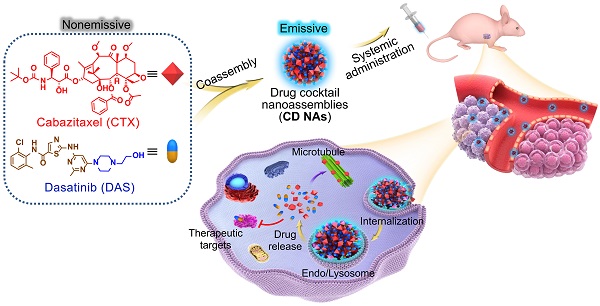
New strategies to fabricate nanomedicines with high translational capacity are urgently desired. Herein, a new class of self-assembled drug cocktails that addresses the multiple challenges of manufacturing clinically useful cancer nanomedicines was reported.
Methods: With the aid of a molecular targeted agent, dasatinib (DAS), cytotoxic cabazitaxel (CTX) forms nanoassemblies (CD NAs) through one-pot process, with nearly quantitative entrapment efficiency and ultrahigh drug loading of up to 100%.
Results: Surprisingly, self-assembled CD NAs show aggregation-induced emission, enabling particle trafficking and drug release in living cells. In preclinical models of human cancer, including a patient-derived melanoma xenograft, CD NAs demonstrated striking therapeutic synergy to produce a durable recession in tumor growth. Impressively, CD NAs alleviated the toxicity of the parent CTX agent and showed negligible immunotoxicity in animals.
Conclusions: Overall, this approach does not require any carrier matrices, offering a scalable and cost-effective methodology to create a new generation of nanomedicines for the safe and efficient delivery of drug combinations.
Keywords: self-assembly, drug cocktail, aggregation-induced emission, nanomedicine, synergistic combination.
 Global reach, higher impact
Global reach, higher impact