13.3
Impact Factor
Theranostics 2021; 11(12):5876-5888. doi:10.7150/thno.55106 This issue Cite
Research Paper
Computed tomography imaging of macrophage phagocytic activity in abdominal aortic aneurysm
1. Cardiovascular Molecular Imaging Laboratory, Section of Cardiovascular Medicine and Yale Cardiovascular Research Center, Yale University School of Medicine, New Haven, CT (USA).
2. Veterans Affairs Connecticut Healthcare System, West Haven, CT (USA).
3. CCMI Electron Microscopy Core Facility, Yale University School of Medicine, New Haven, CT (USA).
Abstract
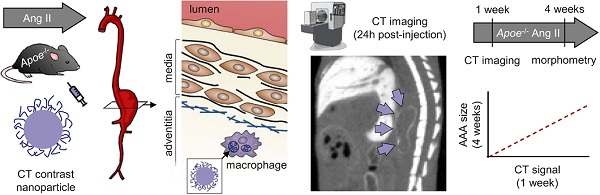
Inflammation plays a major role in the pathogenesis of several vascular pathologies, including abdominal aortic aneurysm (AAA). Evaluating the role of inflammation in AAA pathobiology and potentially outcome in vivo requires non-invasive tools for high-resolution imaging. We investigated the feasibility of X-ray computed tomography (CT) imaging of phagocytic activity using nanoparticle contrast agents to predict AAA outcome.
Methods: Uptake of several nanoparticle CT contrast agents was evaluated in a macrophage cell line. The most promising agent, Exitron nano 12000, was further characterized in vitro and used for subsequent in vivo testing. AAA was induced in Apoe-/- mice through angiotensin II (Ang II) infusion for up to 4 weeks. Nanoparticle biodistribution and uptake in AAA were evaluated by CT imaging in Ang II-infused Apoe-/- mice. After imaging, the aortic tissue was harvested and used from morphometry, transmission electron microscopy and gene expression analysis. A group of Ang II-infused Apoe-/- mice underwent nanoparticle-enhanced CT imaging within the first week of Ang II infusion, and their survival and aortic external diameter were evaluated at 4 weeks to address the value of vessel wall CT enhancement in predicting AAA outcome.
Results: Exitron nano 12000 showed specific uptake in macrophages in vitro. Nanoparticle accumulation was observed by CT imaging in tissues rich in mononuclear phagocytes. Aortic wall enhancement was detectable on delayed CT images following nanoparticle administration and correlated with vessel wall CD68 expression. Transmission electron microscopy ascertained the presence of nanoparticles in AAA adventitial macrophages. Nanoparticle-induced CT enhancement on images obtained within one week of AAA induction was predictive of AAA outcome at 4 weeks.
Conclusions: By establishing the feasibility of CT-based molecular imaging of phagocytic activity in AAA, this study links the inflammatory signal on early time point images to AAA evolution. This readily available technology overcomes an important barrier to cross-sectional, longitudinal and outcome studies, not only in AAA, but also in other cardiovascular pathologies and facilitates the evaluation of modulatory interventions, and ultimately upon clinical translation, patient management.
Keywords: molecular imaging, computed tomography, nanoparticles, inflammation, abdominal aortic aneurysm
 Global reach, higher impact
Global reach, higher impact