13.3
Impact Factor
Theranostics 2021; 11(12):6019-6032. doi:10.7150/thno.59065 This issue Cite
Research Paper
Photosensitizer-driven nanoassemblies of homodimeric prodrug for self-enhancing activation and synergistic chemo-photodynamic therapy
1. Department of Pharmaceutics, Wuya College of Innovation, Shenyang Pharmaceutical University, Shenyang, Liaoning, 110016, P. R. China.
2. Department of Pharmacology, School of Life Science and Biopharmaceutics, Shenyang Pharmaceutical University, Shenyang, Liaoning 110016, PR China.
Abstract
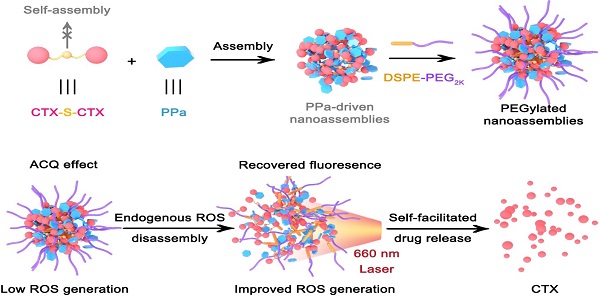
Carrier-free prodrug-nanoassemblies have emerged as promising nanomedicines. In particular, the self-assembled nanoparticles (NPs) composed of homodimeric prodrugs with ultrahigh drug loading have attracted broad attention. However, most homodimeric prodrugs show poor self-assembly ability due to their symmetric structures. Herein, we developed photosensitizer-driven nanoassemblies of homodimeric prodrug for self-enhancing activation and chemo-photodynamic synergistic therapy.
Methods: In this work, a pyropheophorbide a (PPa)-driven nanoassemblies of an oxidation-responsive cabazitaxel homodimer (CTX-S-CTX) was fabricated (pCTX-S-CTX/PPa NPs). The assembly mechanisms, aggregation-caused quenching (ACQ) effect alleviation, singlet oxygen generation, self-enhancing prodrug activation, cellular uptake, intracellular reactive oxygen species (ROS) generation and synergistic cytotoxicity of pCTX-S-CTX/PPa NPs were investigated in vitro. Moreover, the pharmacokinetics, ex vivo biodistribution and in vivo therapeutic efficacy of pCTX-S-CTX/PPa NPs were studied in mice bearing 4T1 tumor.
Results: Interestingly, PPa was found to drive the assembly of CTX-S-CTX, which cannot self-assemble into stable NPs alone. Multiple intermolecular forces were found to be involved in the assembly process. Notably, the nanostructure was destroyed in the presence of endogenous ROS, significantly relieving the ACQ effect of PPa. In turn, ROS generated by PPa under laser irradiation together with the endogenous ROS synergistically promoted prodrug activation. As expected, the nanoassemblies demonstrated potent antitumor activity in a 4T1 breast cancer BALB/c mice xenograft model.
Conclusion: Our findings offer a simple strategy to facilitate the assembly of homodimeric prodrugs and provide an efficient nanoplatform for chemo-photodynamic therapy.
Keywords: Cabazitaxel homodimer, Pyropheophorbide a, Nanoassemblies, Self-enhancing activation, Chemo-photodynamic therapy.
 Global reach, higher impact
Global reach, higher impact