Impact Factor
Theranostics 2021; 11(12):6090-6104. doi:10.7150/thno.56813 This issue Cite
Research Paper
Mechanically induced integrin ligation mediates intracellular calcium signaling with single pulsating cavitation bubbles
1. Department of Mechanical Engineering and Materials Science, Duke University, Durham, NC 27708.
2. Department of Biomedical Engineering, Duke University, Durham, NC 27708.
3. Lee Kong Chian School of Medicine, Nanyang Technological University Singapore, Singapore, 308232.
4. Institute of Molecular and Cell Biology, Agency for Science, Technology and Research (A*STAR), Singapore, Singapore.
Abstract
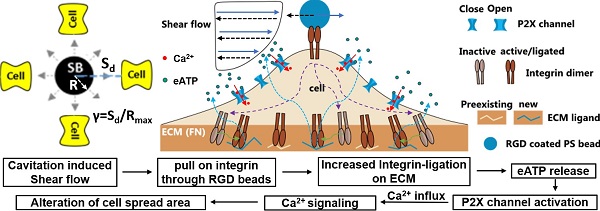
Therapeutic ultrasound or shockwave has shown its great potential to stimulate neural and muscle tissue, where cavitation microbubble induced Ca2+ signaling is believed to play an important role. However, the pertinent mechanisms are unknown, especially at the single-cell level. Particularly, it is still a major challenge to get a comprehensive understanding of the effect of potential mechanosensitive molecular players on the cellular responses, including mechanosensitive ion channels, purinergic signaling and integrin ligation by extracellular matrix.
Methods: Here, laser-induced cavitation microbubble was used to stimulate individual HEK293T cells either genetically knocked out or expressing Piezo1 ion channels with different normalized bubble-cell distance. Ca2+ signaling and potential membrane poration were evaluated with a real-time fluorescence imaging system. Integrin-binding microbeads were attached to the apical surface of the cells at mild cavitation conditions, where the effect of Piezo1, P2X receptors and integrin ligation on single cell intracellular Ca2+ signaling was assessed.
Results: Ca2+ responses were rare at normalized cell-bubble distances that avoided membrane poration, even with overexpression of Piezo1, but could be increased in frequency to 42% of cells by attaching integrin-binding beads. We identified key molecular players in the bead-enhanced Ca2+ response: increased integrin ligation by substrate ECM triggered ATP release and activation of P2X—but not Piezo1—ion channels. The resultant Ca2+ influx caused dynamic changes in cell spread area.
Conclusion: This approach to safely eliciting a Ca2+ response with cavitation microbubbles and the uncovered mechanism by which increased integrin-ligation mediates ATP release and Ca2+ signaling will inform new strategies to stimulate tissues with ultrasound and shockwaves.
Keywords: Intracellular calcium signaling, cavitation bioeffects, Piezo1, integrin ligation, ATP-gated P2X channel.
 Global reach, higher impact
Global reach, higher impact