13.3
Impact Factor
Theranostics 2021; 11(13):6315-6333. doi:10.7150/thno.52843 This issue Cite
Research Paper
CircUbe3a from M2 macrophage-derived small extracellular vesicles mediates myocardial fibrosis after acute myocardial infarction
1. Department of Cardiology, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, China.
2. Department of Cardiology, The Second Affiliated Hospital of Zunyi Medical University, Zunyi 563000, China.
3. Department of Cardiology, Shanghai Institute of Cardiovascular Diseases, Zhongshan Hospital, Fudan University, Shanghai 200032, China.
* These authors contributed equally to this work.
Abstract
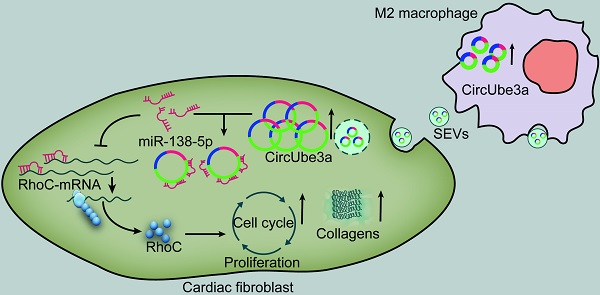
Objective: This study aimed to explore the role of circular RNAs (circRNAs) in M2 macrophage (M2M)-derived small extracellular vesicles (SEVs) in myocardial fibrosis development.
Methods: The regulatory role of M2M-derived extracellular vesicles (EVs) was evaluated in a mouse model of acute myocardial infarction. Immunofluorescence, quantitative real-time PCR (RT-qPCR), nanoparticle tracking analysis, Western blot analysis and electron microscopy were used to identify macrophages, large extracellular vesicles (LEVs) and SEVs. The circRNA expression profiles of M0 macrophages (M0Ms) and M2Ms were determined by microarray analysis. Bioinformatic analysis, cell coculture and cell proliferation assays were performed to investigate the expression, function, and regulatory mechanisms of circUbe3a in vitro. qPCR, RNA immunoprecipitation (RIP), dual-luciferase reporter assays, RNA fluorescence in situ hybridization (RNA-FISH), Western blot analysis and a series of rescue experiments were used to verify the correlation among circUbe3a, miR-138-5p and RhoC.
Results: CircUbe3a from M2M-derived SEVs triggered functional changes in cardiac fibroblasts (CFs). CircUbe3a was synthesized and loaded into SEVs during increased M2M infiltration after myocardial infarction. The fusion of the released SEVs with the plasma membrane likely caused the release of circUbe3a into the cytosol of CFs. Silencing or overexpressing circUbe3a altered CF proliferation, migration, and phenotypic transformation in vitro. We confirmed that circUbe3a plays a crucial role in enhancing functional changes in CFs by sponging miR-138-5p and then translationally repressing RhoC in vitro. In vivo, the addition of M2M-derived SEVs or overexpression of circUbe3a significantly exacerbated myocardial fibrosis after acute myocardial infarction, and these effects were partially abolished by circUbe3a-specific shRNA.
Conclusions: Our findings suggest that M2M-derived circUbe3a-containing SEVs promote the proliferation, migration, and phenotypic transformation of CFs by directly targeting the miR-138-5p/RhoC axis, which may also exacerbate myocardial fibrosis after acute myocardial infarction.
Keywords: CircUbe3a, Small extracellular vesicles, miR-138-5p, Rhoc, Myocardial fibrosis.
 Global reach, higher impact
Global reach, higher impact