Impact Factor
Theranostics 2021; 11(13):6445-6460. doi:10.7150/thno.53637 This issue Cite
Research Paper
Phosphatidylserine-exposing tumor-derived microparticles exacerbate coagulation and cancer cell transendothelial migration in triple-negative breast cancer
1. Department of Ultrasound, The First Hospital, Harbin Medical University, Harbin, China.
2. Department of Gerontology, The First Hospital, Harbin Medical University, Harbin, China.
3. Department of Hematology, The First Hospital, Harbin Medical University, Harbin, China.
4. Department of Research, VA Boston Healthcare System, Boston, MA.
5. Department of Medical Oncology, Dana-Farber Cancer Institute, Boston, MA.
6. Department of Medicine, Brigham and Women's Hospital, and Harvard Medical School, Boston, MA.
* Cong Zhang and Zhuowen Yang are co-first authors of the article.
Abstract
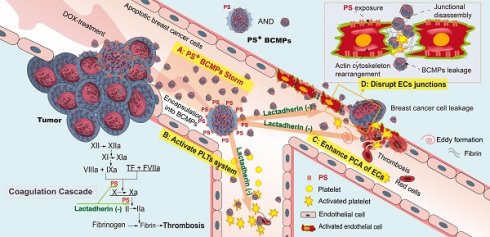
Background: Neoadjuvant chemotherapy is relevant to the formation of thromboembolism and secondary neoplasms in triple-negative breast cancer (TNBC). Chemotherapy-induced breast cancer cell-derived microparticles (BCMPs) may have important thrombogenic and pro-metastatic effects on platelets and endothelium, which may be related to the expression and distribution of phosphatidylserine (PS). However, investigating these interactions is challenging due to technical limitations.
Methods: A study was conducted in 20 healthy individuals and 18 patients who had been recently diagnosed with TNBC and were undergoing neoadjuvant chemotherapy with doxorubicin and cyclophosphamide. BCMPs were isolated from patient blood samples and doxorubicin-treated breast cancer cell lines. Their structure and morphology were studied by electron microscopy and antigen levels were measured by fluorescence-activated cell sorting. In an inhibition assay, isolated BCMPs were pretreated with lactadherin or tissue factor antibodies. Platelets isolated from healthy subjects were treated with BCMPs and coagulation time, fibrin formation, and expression of intrinsic/extrinsic factor Xase (FXa) and thrombin were evaluated. The effects of BCMPs on endothelial thrombogenicity and integrity were assessed by confocal microscopy, electron microscopy, measurement of intrinsic/extrinsic FXa, prothrombinase assay, and transwell permeability assay.
Results: Neoadjuvant chemotherapy significantly increased the expression of PS+ BCMPs in patient plasma. Its expression was associated with a rapid increase in procoagulant activity. Treatment with lactadherin, a PS-binding scavenging molecule, markedly reduced the adhesion of BCMPs and abolished their procoagulant activity, but this was not observed with tissue factor antibody treatment. Intravenous injection of BCMPs in mice induced a significant hypercoagulable state, reducing the extent of plasma fibrinogen and promoting the appearance of new thrombus. Cancer cells incubated with doxorubicin released large numbers of PS+ BCMPs, which stimulated and transformed endothelial cells into a procoagulant phenotype and increased the aggregation and activation of platelets. Moreover, cancer cells exploited this BCMP-induced endothelial leakiness and showed promoted metastasis. Pretreatment with lactadherin increased uptake of both PS+ BCMPs and cancer cells by endothelial cells and limited the transendothelial migration of cancer cells.
Conclusion: Lactadherin, a biosensor that we developed, was used to study the extracellular vesicle distribution of PS, which revealed a novel PS+ BCMPs administrative axis that initiated a local coagulation cascade and facilitated metastatic colonization of circulating cancer cells.
Keywords: Phosphatidylserine, tumor-derived microparticles, procoagulation, transendothelial migration, lactadherin.
 Global reach, higher impact
Global reach, higher impact