Impact Factor
Theranostics 2021; 11(13):6507-6521. doi:10.7150/thno.56367 This issue Cite
Research Paper
Targeted delivery of neural progenitor cell-derived extracellular vesicles for anti-inflammation after cerebral ischemia
1. Department of Neurobiology, Key Laboratory of Human Functional Genomics of Jiangsu, Nanjing Medical University, Nanjing, Jiangsu Province 211166, China.
2. Department of Dermatology, The Affiliated Wuxi No.2 People's Hospital of Nanjing Medical University, Wuxi, Jiangsu Province 214002, China.
3. Department of Rehabilitation Medicine, Jiangsu Shengze Hospital Affiliated to Nanjing Medical University, Suzhou, Jiangsu Province 215228, China.
4. The Department of Oral and Maxillofacial Surgery, The Affiliated Stomatology Hospital of Nanjing Medical University, Nanjing, Jiangsu Province 210029, China.
5. Experimental Therapeutics and Molecular Imaging Lab, Department of Neurology, Massachusetts General Hospital and Harvard Medical School, Boston, MA 02129, United States.
#These authors contributed equally to this work.
Abstract
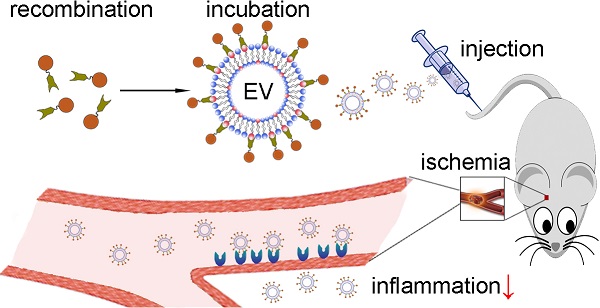
Ischemic stroke remains a major cause of death, and anti-inflammatory strategies hold great promise for preventing major brain injury during reperfusion. In the past decade, stem cell-derived extracellular vesicles (EVs) have emerged as novel therapeutic effectors in immune modulation. However, the intravenous delivery of EVs into the ischemic brain remains a challenge due to poor targeting of unmodified EVs, and the costs of large-scale production of stem cell-derived EVs hinder their clinical application.
Methods: EVs were isolated from a human neural progenitor cell line, and their anti-inflammatory effects were verified in vitro. To attach targeting ligands onto EVs, we generated a recombinant fusion protein containing the arginine-glycine-aspartic acid (RGD)-4C peptide (ACDCRGDCFC) fused to the phosphatidylserine (PS)-binding domains of lactadherin (C1C2), which readily self-associates onto the EV membrane. Subsequently, in a middle cerebral artery occlusion (MCAO) mouse model, the RGD-C1C2-bound EVs (RGD-EV) were intravenously injected through the tail vein, followed by fluorescence imaging and assessment of proinflammatory cytokines expression and microglia activation.
Results: The neural progenitor cell-derived EVs showed intrinsic anti-inflammatory activity. The RGD-EV targeted the lesion region of the ischemic brain after intravenous administration, and resulted in a strong suppression of the inflammatory response. Furthermore, RNA sequencing revealed a set of 7 miRNAs packaged in the EVs inhibited MAPK, an inflammation related pathway.
Conclusion: These results point to a rapid and easy strategy to produce targeting EVs and suggest a potential therapeutic agent for ischemic stroke.
Keywords: extracellular vesicles, exosomes, anti-inflammation, targeted delivery, cerebral ischemia
 Global reach, higher impact
Global reach, higher impact