13.3
Impact Factor
Theranostics 2021; 11(13):6542-6559. doi:10.7150/thno.54589 This issue Cite
Research Paper
Longitudinal evaluation of a novel BChE PET tracer as an early in vivo biomarker in the brain of a mouse model for Alzheimer disease
1. University of Ljubljana, Faculty of Chemistry and Chemical Technology, Večna pot 113, 1000 Ljubljana, Slovenia
2. CIC biomaGUNE, Basque Research and Technology Alliance (BRTA), Paseo Miramon 182, 20014, San Sebastian, Spain.
3. Laboratory of Neuroimaging and biomarkers of inflammation, Achucarro Basque Center for Neuroscience, Science Park UPV/EHU, 48940 Leioa, Spain
4. Multiple Sclerosis Unit, Biodonostia Health Institute, Donostia-San Sebastián, Spain
5. Department of Nuclear Medicine, University Hospital of Araba (HUA), 01009 Vitoria-Gasteiz, Spain.
6. Department of Surgery Radiology and Physical Medicine, Faculty of Medicine, University of the Basque Country, UPV/EHU,01009 Vitoria-Gasteiz, Spain
7. IKERBASQUE, Basque Foundation for Science, 48013 Bilbao, Spain
8. Departament de Biomedicina, Facultat de Medicina i Ciències de la Salut, Institut de Neurociències. Barcelona, Spain.
9. Institut d'Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS), 08036 Barcelona, Spain.
10. Centro de Investigación Biomédica en Red sobre Enfermedades Neurodegenerativas (CIBERNED), 28031 Madrid, Spain.
11. Production and Validation Center of Advanced Therapies (Creatio), Faculty of Medicine and Health Science, University of Barcelona, 08036 Barcelona, Spain.
12. Department of Brain Sciences, Imperial College London, Hammersmith Hospital, Du Cane Road, London W12 0NN.
13. Department of Neuroscience, Faculty of Medicine and Nursery, University of the Basque Country UPV/EHU and Achucarro Basque Center for Neuroscience, Barrio Sarriena S/N, 48940 Leioa, Spain.
14. University of Ljubljana, Faculty of Pharmacy, Aškerčeva cesta 7, 1000 Ljubljana, Slovenia.
15. Universidad de Buenos Aires, Consejo Nacional de Investigaciones Científicas y Técnicas, and Instituto de Química y Fisicoquímica Biológicas, Facultad de Farmacia y Bioquímica, Universidad de Buenos Aires, Junín 956, C1113AAD Buenos Aires, Argentina
16. Centro de Investigación Biomédica en Red - Enfermedades Respiratorias (CIBERES).
Abstract
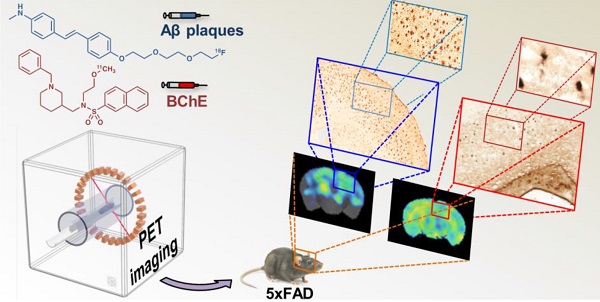
Purpose: The increase in butyrylcholinesterase (BChE) activity in the brain of Alzheimer disease (AD) patients and animal models of AD position this enzyme as a potential biomarker of the disease. However, the information on the ability of BChE to serve as AD biomarker is contradicting, also due to scarce longitudinal studies of BChE activity abundance. Here, we report 11C-labeling, in vivo stability, biodistribution, and longitudinal study on BChE abundance in the brains of control and 5xFAD (AD model) animals, using a potent BChE selective inhibitor, [11C]4, and positron emission tomography (PET) in combination with computerised tomography (CT). We correlate the results with in vivo amyloid beta (Aβ) deposition, longitudinally assessed by [18F]florbetaben-PET imaging.
Methods: [11C]4 was radiolabelled through 11C-methylation. Metabolism studies were performed on blood and brain samples of female wild type (WT) mice. Biodistribution studies were performed in female WT mice using dynamic PET-CT imaging. Specific binding was demonstrated by ex vivo and in vivo PET imaging blocking studies in female WT and 5xFAD mice at the age of 7 months. Longitudinal PET imaging of BChE was conducted in female 5xFAD mice at 4, 6, 8, 10 and 12 months of age and compared to age-matched control animals. Additionally, Aβ plaque distribution was assessed in the same mice using [18F]florbetaben at the ages of 2, 5, 7 and 11 months. The results were validated by ex vivo staining of BChE at 4, 8, and 12 months and Aβ at 12 months on brain samples.
Results: [11C]4 was produced in sufficient radiochemical yield and molar activity for the use in PET imaging. Metabolism and biodistribution studies confirmed sufficient stability in vivo, the ability of [11C]4 to cross the blood brain barrier (BBB) and rapid washout from the brain. Blocking studies confirmed specificity of the binding. Longitudinal PET studies showed increased levels of BChE in the cerebral cortex, hippocampus, striatum, thalamus, cerebellum and brain stem in aged AD mice compared to WT littermates. [18F]Florbetaben-PET imaging showed similar trend of Aβ plaques accumulation in the cerebral cortex and the hippocampus of AD animals as the one observed for BChE at ages 4 to 8 months. Contrarily to the results obtained by ex vivo staining, lower abundance of BChE was observed in vivo at 10 and 12 months than at 8 months of age.
Conclusions: The BChE inhibitor [11C]4 crosses the BBB and is quickly washed out of the brain of WT mice. Comparison between AD and WT mice shows accumulation of the radiotracer in the AD-affected areas of the brain over time during the early disease progression. The results correspond well with Aβ accumulation, suggesting that BChE is a promising early biomarker for incipient AD.
Keywords: Butyrylcholinesterase, PET, Positron Emission Tomography, Alzheimer Disease, Amyloid Beta.
 Global reach, higher impact
Global reach, higher impact