13.3
Impact Factor
Theranostics 2021; 11(13):6560-6572. doi:10.7150/thno.55609 This issue Cite
Research Paper
Organ-specific cholesterol metabolic aberration fuels liver metastasis of colorectal cancer
1. Department of General Surgery, Huashan Hospital & Cancer Metastasis Institute & Institutes of Biomedical Sciences, Fudan University, Shanghai, 200040, China.
2. Department of Pathology, Huashan Hospital, Fudan University, Shanghai, 200040, China.
*Present address: Shanghai Institute of Nutrition and Health, Chinese Academy of Sciences, Shanghai, 200031, China.
#These authors contributed equally to this work.
Abstract
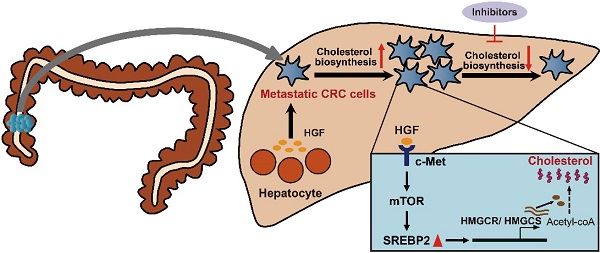
Rationale: Metastasis, the development of secondary malignant growth at a distance from a primary tumor, is the main cause of cancer-associated death. However, little is known about how metastatic cancer cells adapt to and colonize in the new organ environment. Here we sought to investigate the functional mechanism of cholesterol metabolic aberration in colorectal carcinoma (CRC) liver metastasis.
Methods: The expression of cholesterol metabolism-related genes in primary colorectal tumors (PT) and paired liver metastases (LM) were examined by RT-PCR. The role of SREBP2-dependent cholesterol biosynthesis pathway in cell growth and CRC liver metastasis were determined by SREBP2 silencing in CRC cell lines and experimental metastasis models including, intra-splenic injection models and liver orthotropic injection model. Growth factors treatment and co-culture experiment were performed to reveal the mechanism underlying the up-regulation of SREBP2 in CRC liver metastases. The in vivo efficacy of inhibition of cholesterol biosynthesis pathway by betulin or simvastatin were evaluated in experimental metastasis models.
Results: In the present study, we identify a colorectal cancer (CRC) liver metastasis-specific cholesterol metabolic pathway involving the activation of SREBP2-dependent cholesterol biosynthesis, which is required for the colonization and growth of metastatic CRC cells in the liver. Inhibiting this cholesterol biosynthesis pathway suppresses CRC liver metastasis. Mechanically, hepatocyte growth factor (HGF) from liver environment activates SREBP2-dependent cholesterol biosynthesis pathway by activating c-Met/PI3K/AKT/mTOR axis in CRC cells.
Conclusion: Our findings support the notion that CRC liver metastases show a specific cholesterol metabolic aberration. Targeting this cholesterol biosynthesis pathway could be a promising treatment for CRC liver metastasis.
Keywords: colorectal cancer, liver metastasis, SREBP2, cholesterol biosynthesis, HGF
 Global reach, higher impact
Global reach, higher impact