13.3
Impact Factor
Theranostics 2021; 11(13):6632-6643. doi:10.7150/thno.57706 This issue Cite
Research Paper
EGFR inhibition blocks cancer stem cell clustering and lung metastasis of triple negative breast cancer
1. Department of Pharmacology, Northwestern University Feinberg School of Medicine, Chicago, IL, USA.
2. Department of Toxicology and Cancer Biology, University of Kentucky, Lexington, KY, USA.
3. Department of Pharmacology, Case Western Reserve University, Cleveland, OH, USA.
4. The Ben May Department for Cancer Research, the University of Chicago, Chicago, IL, USA.
5. Laboratory of Functional Biology, Graduate School of Biostudies, Kyoto University, Kyoto, Japan.
6. Drikskill Graduate Program in Life Sciences, Northwestern University Feinberg School of Medicine, Chicago, IL, USA.
7. Department of Medicine, the Division of Hematology and Oncology, Northwestern University Feinberg School of Medicine, Chicago, IL, USA.
8. Lurie Comprehensive Cancer Center, Northwestern University Feinberg School of Medicine, Chicago, IL, USA.
9. Department of Pathology and Case Comprehensive Cancer Center, Case Western Reserve University, Cleveland, OH, USA.
*These authors contributed equally to the work.
Abstract
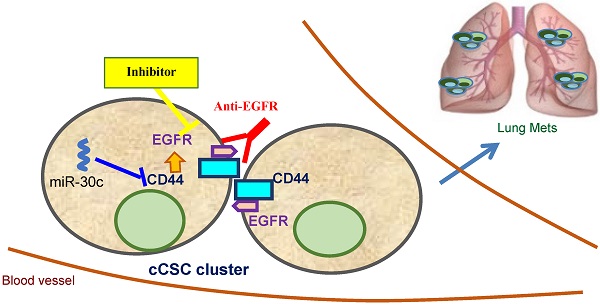
Triple-negative breast cancer (TNBC) is one of the most aggressive and metastatic breast cancer subtypes lacking targeted therapy. Our recent work demonstrated that circulating tumor cell (CTC) clusters and polyclonal metastasis of TNBC are driven by aggregation of CD44+ cancer stem cells (CSC) and associated with an unfavorable prognosis, such as low overall survival. However, there is no existing therapeutic that can specifically block CTC or CSC cluster formation.
Methods: Using patient-derived xenograft (PDX) models, we established an ex vivo tumor cell clustering assay for a pilot screening of blockade antibodies. After identifying EGFR as a target candidate, we modulated the gene expression and inhibited its kinase activity to determine its functional importance in tumor cell clustering and therapeutic inhibition of lung metastasis. We also examined the molecular regulation network of EGFR and a potential connection to CSC marker CD44 and microRNAs, which regulate CTC clustering.
Results: We report here that EGFR inhibition successfully blocks circulating CSC (cCSC) clustering and lung metastasis of TNBC. EGFR enhances CD44-mediated tumor cell aggregation and CD44 stabilizes EGFR. Importantly, blocking EGFR by a novel anti-EGFR monoclonal antibody (clone LA1) effectively blocked cell aggregation in vitro and reduced lung metastasis in vivo. Furthermore, our data demonstrated that the tumor suppressor microRNA-30c serves as another negative regulator of cCSC clustering and lung metastasis by targeting CD44 as well as its downstream effector EGFR.
Conclusion: Our studies identify a novel anti-EGFR therapeutic strategy to inhibit cCSC aggregation and therefore abolish cCSC cluster-mediated metastasis of TNBC.
Keywords: cancer stem cells, metastasis, EGFR, circulating tumor cells, CTC cluster, circulating cancer stem cell
 Global reach, higher impact
Global reach, higher impact