13.3
Impact Factor
Theranostics 2021; 11(15):7247-7261. doi:10.7150/thno.58468 This issue Cite
Research Paper
Major vault protein (MVP) negatively regulates osteoclastogenesis via calcineurin-NFATc1 pathway inhibition
1. Jiangsu Key Laboratory of Oral Diseases, Nanjing Medical University, 140 Hanzhong Road, Nanjing 210029, China.
2. Department of Pathophysiology, Key Laboratory of Cardiovascular Disease and Molecular Intervention, Nanjing Medical University, Nanjing 211166, China.
3. Department of Medical Oncology, Erasmus MC Cancer Institute, Erasmus University Medical Center, Rotterdam, Netherland.
4. Suzhou Huaxia Stomatology Hospital affiliated to Suzhou Vocational Health College, Suzhou 215009, China.
*These authors contributed equally to this work.
Abstract
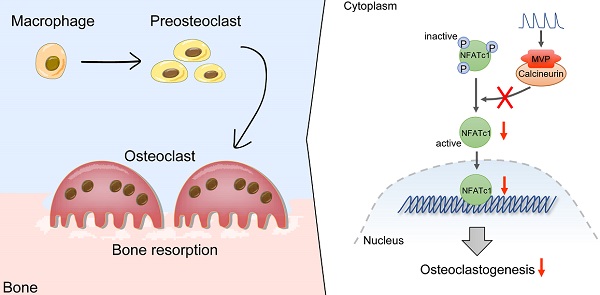
Rationale: Bone homeostasis is maintained by a balanced interplay of osteoblasts and osteoclasts. Osteoclasts are derived from monocyte/macrophage lineage. Major vault protein (MVP) is known to promote apoptosis and prevent metabolic diseases in macrophage. However, whether MVP is involved in osteoclastogenesis is unknown. Here, we identified an important function of MVP as a negative regulator of osteoclastogenesis and its therapeutic potential in preventing bone loss.
Methods: Expression of MVP in osteoclasts was investigated in human tumor tissues with immunohistochemical staining. Next, we generated total body (Mvp-/-) and monocyte-specific (Mvpf/fLyz2-Cre) MVP gene knockout mice to observe bone phenotype and osteoclastogenesis using micro-CT and bone histomorphometry. Moreover, we examined the effects of MVP on osteoclast differentiation, bone resorption, NFATc1 activation and calcium oscillations in vitro. Finally, we explored the clinical potential of targeting MVP in two osteoporosis mouse models and used an adeno-associated virus (AAV) gene to overexpress MVP locally in mice.
Results: We found that Mvp-/- and Mvpf/fLyz2-Cre mice both exhibited osteoporosis-like phenotypes. MVP-deficiency also enhanced calcineurin-NFATc1 signaling and promoted NFATc1 activity, which led to enhanced osteoclastogenesis and bone resorption. Calcineurin inhibition using the small molecule inhibitor FK506 corrected the enhanced osteoclastogenesis in Mvpf/fLyz2-Cre group. Additionally, MVP reexpression in Mvpf/fLyz2-Cre group rescued calcineurin expression. MVP overexpression in wild-type mice prevented pathologic bone loss in mouse models of ovariectomized (OVX) and calvaria-adjacent lipopolysaccharide (LPS)-injected.
Conclusions: Our data suggested that MVP negatively regulates osteoclast differentiation and bone resorption via inhibition of calcineurin-NFATc1 signaling. In osteoclast-related bone diseases such as osteoporosis, manipulation of MVP activity may be an attractive therapeutic target.
Keywords: MVP, osteoclast, NFATc1, calcineurin, osteoporosis
 Global reach, higher impact
Global reach, higher impact