13.3
Impact Factor
Theranostics 2021; 11(17):8270-8282. doi:10.7150/thno.60758 This issue Cite
Research Paper
Cell membrane inspired nano-shell enabling long-acting Glucose Oxidase for Melanoma starvation therapy via microneedles-based percutaneous delivery
1. Xiangya School of Pharmaceutical Sciences, Central South University, Changsha, Hunan, 410013, China
2. Department of Pathology, School of basic medicine, Central South University, Changsha, Hunan, 410013, China
Abstract
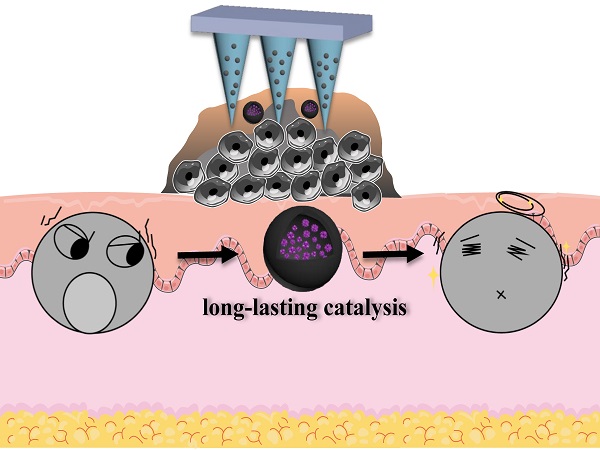
Rationale: Glucose oxidase (GOx) has gained tremendous research interest recently as a glucose-consuming enzyme for tumor starvation therapy, while its in vivo applications are strictly limited by rapid deactivation, as well as side effects of non-specific catalysis.
Methods: To address these issues, here we report a protective nano-shell to encapsule GOx for localized melanoma therapy delivered by dissolving microneedles (MNs). Inspired by cell membrane that separates and protects cell organelles and components from outside environment while selectively ingesting nutrition sources, we designed polydopamine (PDA)-structured nano-shell to allow free transportation of glucose for catalytic reaction, while impede the penetration of GOx, proteinase, and other GOx-deactivating macromolecules across the shell membrane.
Results: GOx was well protected in core layer with persistent catalytic activity for at least 6 d under various biological matrixes (e.g., PBS, serum, and cell lysate) and surviving different harsh conditions (e.g., acid/base treatments, and proteinase-induced degradation). Such long-acting nano-catalyst can be easily integrated into MNs as topical delivery carrier for effective glucose consumption in melanoma tissue, achieving significant tumor growth inhibition via starvation therapy with minimized side effects as compared to systemic administration.
Conclusion: This work provides an elegant platform for in vivo delivery of GOx, and our cell-mimicking nano-system can also be applied for other enzyme-based therapeutics.
Keywords: Skin cancer, starvation therapy, catalysis, metal organic frameworks, polydopamine
 Global reach, higher impact
Global reach, higher impact