13.3
Impact Factor
Theranostics 2021; 11(17):8301-8321. doi:10.7150/thno.61214 This issue Cite
Research Paper
Microenvironment Activatable Nanoprodrug Based on Gripper-like Cyclic Phenylboronic Acid to Precisely and Effectively Alleviate Drug-induced Hepatitis
1. Personalized Drug Therapy Key Laboratory of Sichuan Province, Department of Pharmacy, Sichuan Academy of Medical Science & Sichuan Provincial People's Hospital, School of Medicine, University of Electronic Science and Technology of China, Chengdu 610072, China
2. College of Pharmacy, Southwest Minzu University, Chengdu 610000, China
* These authors contributed equally to this work
Abstract
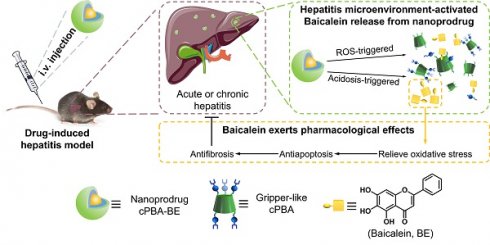
Drug-induced hepatitis (DIH), which seriously interferes with disease treatment, is one of the most common reasons for termination of new drugs during preclinical studies or post-marketing surveillance. Although antioxidants and anti-inflammatory agents are promising, their nonspecific distribution and insolubility limit their application. Therefore, precise drug release at the disease site is an important way to alleviate DIH and avoid side effects.
Methods: A gripper-like hydrophilic cyclic phenylboronic acid (cPBA) was synthesized and a nanoprodrug (cPBA-BE) was established by coupling cPBA with hydrophobic baicalein (BE). The stimuli-responsive release properties and therapeutic effect of cPBA-BE on drug-injured hepatocyte were investigated. The biodistribution and therapeutic effect of cPBA-BE both in acetaminophen-induced acute hepatitis model and rifampicin-induced chronic hepatitis model were further evaluated.
Results: cPBA-BE conjugate could self-assemble into nanoprodrug with cPBA as the hydrophilic external layer and BE as the hydrophobic core. In HepaRG cells, cPBA-BE showed stronger cellular uptake. Due to the H2O2- and acid-sensitivity, cPBA-BE could achieve adequate BE release, significantly resist the depletion of GSH, mitochondrial dysfunction, downregulation of inflammation and cell apoptosis in the acetaminophen injured HepaRG cells. Biodistribution showed that cPBA-BE specifically increased the concentration of BE in the liver of DIH mice. cPBA-BE could alleviate acetaminophen-induced acute hepatitis or rifampicin-induced chronic hepatitis more effectively through relieving the oxidative stress, inflammation and block the neutrophil infiltration in liver.
Conclusions: cPBA is expected to be a good platform for constructing injectable nanoprodrug with both H2O2 and pH-responsive properties by coupling a wide range of drugs containing o-diol. In this study, the nanoprodrug cPBA-BE was determined to be effective for alleviating the DIH.
Keywords: Self-assembly, Microenvironment-activatable, Injectable-nanoprodrug, Inflammation, Targeted therapy
 Global reach, higher impact
Global reach, higher impact