Impact Factor
Theranostics 2021; 11(20):9775-9790. doi:10.7150/thno.61677 This issue Cite
Research Paper
Identification of three tumor antigens and immune subtypes for mRNA vaccine development in diffuse glioma
1. Department of Neurosurgery, Xiangya Hospital, Central South University, Changsha, Hunan Province, 410008, China.
2. Cancer Research Institute, School of Basic Medical Science, Central South University, Changsha, China.
3. Changsha Kexin Cancer Hospital, Changsha, Hunan 410205, China.
4. The NHC Key Laboratory of Carcinogenesis and the Key Laboratory of Carcinogenesis and Cancer Invasion of the Chinese Ministry of Education, Xiangya Hospital, Central South University, Changsha, China.
Abstract
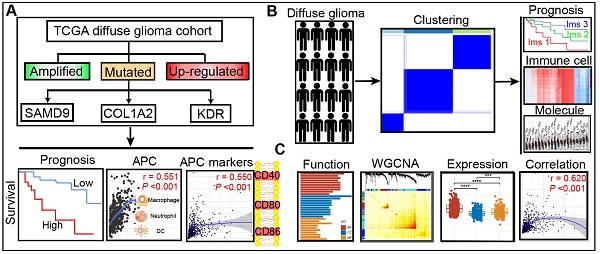
Rationale: Diffuse glioma patients have high mortality and recurrence despite multimodal therapies. This study aims to identify the potential tumor antigens for mRNA vaccines and subtypes suitable for the immunotherapy of patients with diffuse glioma.
Methods: Gene expression profiles and corresponding clinical information were obtained from the Chinese Glioma Genome Atlas (CGGA) and the Cancer Genome Atlas (TCGA) databases. Genetic alterations were extracted from cBioPortal. Differential gene analysis, survival analysis, correlation analysis, consensus clustering analysis, and immune cell infiltration analysis were conducted based on the various databases. Finally, the hub genes, the modules related to tumor antigens, and the immune subtypes were identified using WGCNA method.
Results: Three over-expressed, amplified, and mutated tumor antigens, including KDR, COL1A2, and SAMD9, were associated with clinical outcomes. The expression of the three genes had a positive correlation with the abundance of antigen-presenting cells (APCs) and APC marker expression. Subsequently, three immune subtypes (Ims1, Ims2, and Ims3) were distinguished in the TCGA cohort, which exhibited distinct molecular, cellular, and clinical characteristics consistent with the CGGA cohort. Diffuse gliomas with subtype Ims1 were more malignant with immunosuppressive phenotypes and more associated with poor prognosis than the other two subtypes. The three antigens and the immune checkpoints were differentially expressed among the three immune subtypes. Finally, functional enrichment analysis of the genes related to tumor antigens and immune subtypes suggested that they are enriched in many immune-associated processes.
Conclusions: KDR, COL1A2, and SAMD9 are potential antigens for developing mRNA vaccines against diffuse glioma. The results suggest that immunotherapy targeting these three antigens is more suitable for patients with subtype Ims1. This study provides insights into immunotherapy for diffuse glioma.
Keywords: diffuse glioma, tumor antigen, immune subtypes, mRNA vaccine, tumor immune infiltration
 Global reach, higher impact
Global reach, higher impact