Impact Factor
Theranostics 2021; 11(20):9791-9804. doi:10.7150/thno.64601 This issue Cite
Research Paper
CLEC-2-dependent platelet subendothelial accumulation by flow disturbance contributes to atherogenesis in mice
1. Cyrus Tang Hematology Center, Cyrus Tang Medical Institute, Soochow University, Suzhou, China.
2. Collaborative Innovation Center of Hematology of Jiangsu Province, Soochow University, Suzhou, China.
3. Suzhou Key Lab for Thrombosis and Vascular Biology, Soochow University, Suzhou, China.
4. Cardiovascular Biology Research Program, Oklahoma Medical Research Foundation, Oklahoma City, OK, USA.
*These authors contributed equally to this work.
Abstract
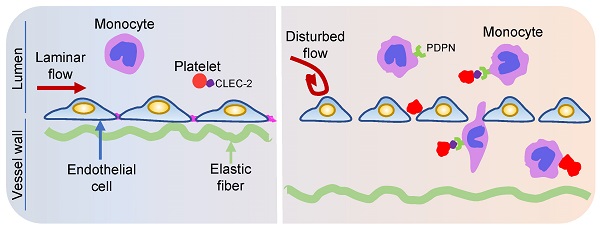
Rationale: Platelets play an essential role in atherosclerosis, but the underlying mechanisms remain to be addressed. This study is to investigate the role of platelets in d-flow induced vascular inflammation and the underlying mechanism.
Methods: We established a disturbed blood flow (d-flow) model by partial carotid ligation (PCL) surgery using atherosclerosis-susceptible mice and wild-type mice to observe the d-flow induced platelet accumulation in the subendothelium or in the plaque by immunostaining or transmission electron microscopy. The mechanism of platelet subendothelial accumulation was further explored by specific gene knockout mice.
Results: We observed presence of platelets in atherosclerotic plaques either in the atheroprone area of aortic arch or in carotid artery with d-flow using Ldlr-/- or ApoE-/- mice on high fat diet. Immunostaining showed the subendothelial accumulation of circulating platelets by d-flow in vivo. Transmission electron microscopy demonstrated the accumulation of platelets associated with monocytes in the subendothelial spaces. The subendothelial accumulation of platelet-monocyte/macrophage aggregates reached peak values at 2 days after PCL. In examining the molecules that may mediate the platelet entry, we found that deletion of platelet C-type lectin-like receptor 2 (CLEC-2) reduced the subendothelial accumulation of platelets and monocytes/macrophages by d-flow, and ameliorated plaque formation in Ldlr-/- mice on high fat diet. Supportively, CLEC-2 deficient platelets diminished their promoting effect on the migration of mouse monocyte/macrophage cell line RAW264.7. Moreover, monocyte podoplanin (PDPN), the only ligand of CLEC-2, was upregulated by d-flow, and the myeloid-specific PDPN deletion mitigated the subendothelial accumulation of platelets and monocytes/macrophages.
Conclusions: Our results reveal a new CLEC-2-dependent platelet subendothelial accumulation in response to d-flow to regulate vascular inflammation.
Keywords: Atherosclerosis, CLEC-2, Disturbed flow, Platelets
 Global reach, higher impact
Global reach, higher impact