Impact Factor
Theranostics 2022; 12(5):2080-2094. doi:10.7150/thno.69444 This issue Cite
Research Paper
HDAC5 modulates PD-L1 expression and cancer immunity via p65 deacetylation in pancreatic cancer
1. Department of Pancreatic Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022 China.
2. Sino-German Laboratory of Personalized Medicine for Pancreatic Cancer, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022 China.
3. Department of Urology, The Second Xiangya Hospital, Central South University, Changsha, Hunan, 410011, China.
4. Uro-Oncology Institute of Central South University, Changsha, Hunan, 410011, China.
5. Cancer Center, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022 China.
6. Clinical Immunology Translational Medicine Key Laboratory of Sichuan Province, Organ Transplant Center, Sichuan Provincial People's Hospital, University of Electronic Science and Technology of China, Chengdu, 611731 China.
*These authors contributed equally to this work.
Abstract
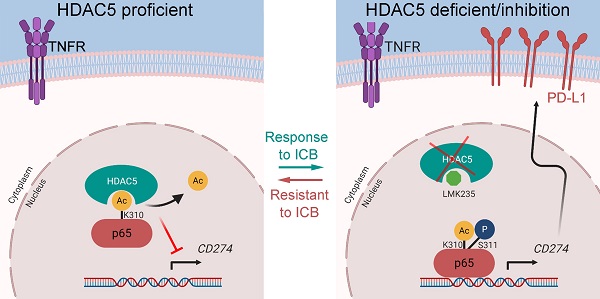
Rationale: Pancreatic ductal adenocarcinoma (PDAC) is a lethal disease with a dismal 5-year survival less than 10%. Most patients with PDAC exhibit poor response to single-agent immunotherapy. Multimodal therapies targeting mechanisms of resistance to immunotherapy are urgently needed. We found that the class IIa histone deacetylase (HDAC) member, HDAC5 is downregulated in multiple solid tumors and its level were associated with favorable prognosis in PDAC patients. Upregulated genes in patients harboring HDAC5 deletions were enriched in adaptive immune responses and lymphocyte-mediated immunity in The Cancer Genome Atlas (TCGA) pancreatic cancer dataset.
Methods: Tissue microarray of pancreatic cancer were used to analysis the correlation between HDAC5 and PD-L1. RNA-seq, transcription factor motif analysis, drug screening and molecular biology assays were performed to identify the mechanism of HDAC5's repression on PD-L1. Allografts of pancreatic cancer in mouse were applied to test the efficiency of HDAC5 inhibition and anti-PD1 co-treatment.
Results: HDAC5 regulated PD-L1 expression by directly interacting with NF-κB p65; this interaction was suppressed by p65 phosphorylation at serine-311. Additionally, HDAC5 diminished p65 acetylation at lysine-310, which is essential for the transcriptional activity of p65. Importantly, we demonstrated that HDAC5 silencing or inhibition sensitized PDAC tumors to immune checkpoint blockade (ICB) therapy in syngeneic mouse model and KPC mouse derived PDAC model.
Conclusion: Our findings revealed a previously unknown role of HDAC5 in regulating the NF-κB signaling pathway and antitumor immune responses. These findings provide a strong rationale for augment the antitumor effects of ICB in immunotherapy-resistant PDAC by inhibiting HDAC5.
Keywords: Pancreatic cancer, Immunotherapy, PD-L1, HDAC5, NF-κB
 Global reach, higher impact
Global reach, higher impact