Impact Factor
Theranostics 2022; 12(5):2232-2247. doi:10.7150/thno.69320 This issue Cite
Research Paper
Interleukin 33-mediated inhibition of A-type K+ channels induces sensory neuronal hyperexcitability and nociceptive behaviors in mice
1. Department of Geriatrics & Clinical Research Center of Neurological Disease, the Second Affiliated Hospital of Soochow University, Suzhou 215004, P.R. China.
2. Department of Physiology and Neurobiology, Medical College of Soochow University, Suzhou 215123, P.R. China.
3. Institute of Regenerative Biology and Medicine, Helmholtz Zentrum München, Munich 81377, Germany.
4. Department of Neurology and Clinical Research Center of Neurological Disease, the Second Affiliated Hospital of Soochow University, Suzhou 215004, P.R. China.
5. Centre for Ion Channelopathy, Soochow University, Suzhou 215123, P.R. China.
6. Jiangsu Key Laboratory of Neuropsychiatric Diseases, Soochow University, Suzhou 215123, P.R. China.
#These authors contributed equally to this work.
Abstract
Background: Interleukin-33 (IL-33) has been implicated in nociceptive pain behaviors. However, the underlying molecular and cellular mechanisms remain unclear.
Methods: Using electrophysiological recording, immunoblot analysis, immunofluorescence labeling, reverse transcription-PCR, siRNA-mediated knockdown approach and behavior tests, we determined the role of IL-33 in regulating sensory neuronal excitability and pain sensitivity mediated by A-type K+ channels.
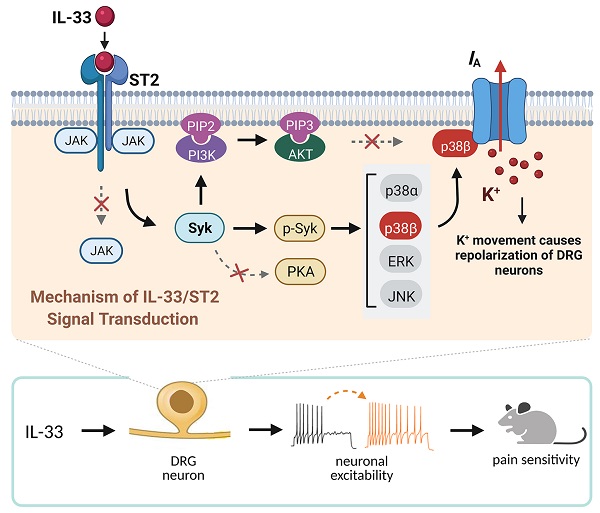
Results: IL-33 decreased A-type transient outward K+ currents (IA) in small-sized DRG neurons in a concentration-dependent manner, whereas the delayed rectifier currents (IDR) remained unaffected. This IL-33-induced IA decrease was dependent on suppression of the tumorigenicity 2 (ST2) receptor and was associated with a hyperpolarizing shift in the steady-state inactivation. Antagonism of Syk abrogated the IL-33-induced IA response, while inhibition of JAK2 and PKA elicited no such effect. Exposure of DRG cells to IL-33 increased the activity of Akt, but surprisingly, neither Akt nor PI3K influenced the IL-33-induced IA response. IL-33 increased the level of phosphorylated p38 mitogen-activated protein kinase (MAPK). Chemical inhibition of p38 and genetic siRNA knockdown of p38 beta (p38β), but not p38α, abrogated the IA response induced by IL-33. Moreover, IL-33 increased neuronal excitability of DRG neurons and facilitated peripheral pain sensitivity in mice; both of these effects were occluded by IA blockade.
Conclusions: Our present study reveals a novel mechanism by which IL-33/ST2 suppresses IA via a Syk-dependent p38β signaling pathway. This mechanism thereby increases DRG neuronal excitability and pain sensitivity in mice. Targeting IL-33/ST2-mediated p38β signaling may represent a therapeutic approach to ameliorate pain behaviors.
Keywords: interleukin 33, A-type K+ channel, dorsal root ganglion neurons, pain
 Global reach, higher impact
Global reach, higher impact