Impact Factor
Theranostics 2022; 12(5):2248-2265. doi:10.7150/thno.65916 This issue Cite
Research Paper
Hepatic RACK1 deficiency protects against fulminant hepatitis through myeloid-derived suppressor cells
1. Beijing Institute of Basic Medical Sciences, Beijing 100850, China.
2. Chinese Institute for Brain Research, Beijing 102206, China.
*These authors contributed equally to this work.
Abstract
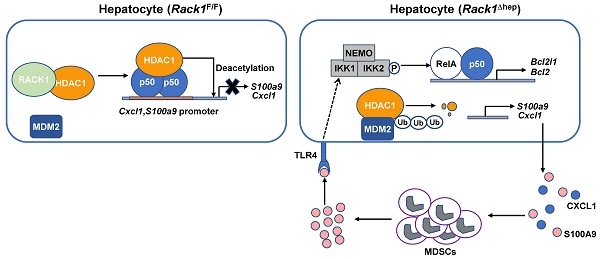
Fulminant hepatitis (FH) is a life-threatening disease with partially understood pathogenesis. It has been demonstrated that myeloid-derived suppressor cells (MDSCs) are recruited into the liver during this process, and their augmented accumulation by various strategies protects against liver injury. However, the underlying mechanism(s) remain elusive. Receptor for activated C kinase 1 (RACK1), a multi-functional scaffold protein, is highly expressed in normal liver and has been implicated in liver physiology and diseases, but the in vivo role of hepatic RACK1 in FH remains unknown.
Methods: Survival curves and liver damage were monitored to investigate the in vivo role of hepatic RACK1 in FH. The liver microenvironment was explored by microarray-based transcriptome analysis, flow cytometry, immunoblotting, and immunohistochemistry. MDSCs were identified with phenotypic and functional characteristics. Functional antibodies were used to target MDSCs. Co-culture techniques were used to study the underlying mechanism(s) of protection. The interaction of RACK1 with histone deacetylase 1 (HDAC1) and the consequent effects on HDAC1 ubiquitination were analyzed. Ectopic expression of HDAC1 with recombinant adeno-associated virus serotype 8 was conducted to confirm the role of HDAC1 in the protective effects of hepatic RACK1 deficiency against FH. Post-translational modifications of RACK1 were also investigated during the induction of FH.
Results: Liver-specific RACK1 deficiency rendered mice resistant to FH. RACK1-deficient livers exhibited high basal levels of chemokine (C-X-C motif) ligand 1 (CXCL1) and S100 calcium-binding protein A9 (S100A9), associated with MDSC accumulation under steady-state conditions. Targeting MDSCs with an antibody against either Gr1 or DR5 abrogated the protective effects of liver-specific RACK1 deficiency. Accumulated MDSCs inhibited inflammatory cytokine production from macrophages and enhanced IκB kinase (IKK)/NF-κB pathway activation in hepatocytes. Further investigation revealed that RACK1 maintained HDAC1 protein level in hepatocytes by direct binding, thereby controlling histone H3K9 and H3K27 acetylation at the Cxcl1 and S100a9 promoters. Ectopic expression of HDAC1 in livers with RACK1 deficiency partially reversed the augmented Cxcl1/S100a9 → MDSCs → IKK/NF-κB axis. During FH induction, RACK1 was phosphorylated at serine 110, enhancing its binding to ubiquitin-conjugating enzyme E2T and promoting its ubiquitination and degradation.
Conclusion: Liver-specific RACK1 deficiency protects against FH through accelerated HDAC1 degradation and the consequent CXCL1/S100A9 upregulation and MDSC accumulation.
Keywords: Fulminant Hepatitis, RACK1, HDAC1, Ubiquitination, MDSCs
 Global reach, higher impact
Global reach, higher impact