Impact Factor
Theranostics 2022; 12(5):2335-2350. doi:10.7150/thno.68715 This issue Cite
Research Paper
A low molecular weight multifunctional theranostic molecule for the treatment of prostate cancer
1. Department of Biomedical Engineering, Case Western Reserve University, 11100 Euclid Ave, Wearn Building B-49, Cleveland, OH, USA, 44106.
2. Department of Radiology, 11100 Euclid Ave, Wearn Building B-44, Case Western Reserve University, Cleveland, OH, USA, 44106.
3. Department of Chemistry, Case Western Reserve University, 2080 Adelbert Rd, Cleveland, OH, USA, 44106.
4. Department of Population and Quantitative Health Sciences, Case Western Reserve University, 2103 Cornell Rd, Cleveland, OH, USA, 44106.
5. Department of Nutritional Biochemistry and Metabolism, 2109 Adelbert Rd, Cleveland, OH, USA, 44106.
Abstract
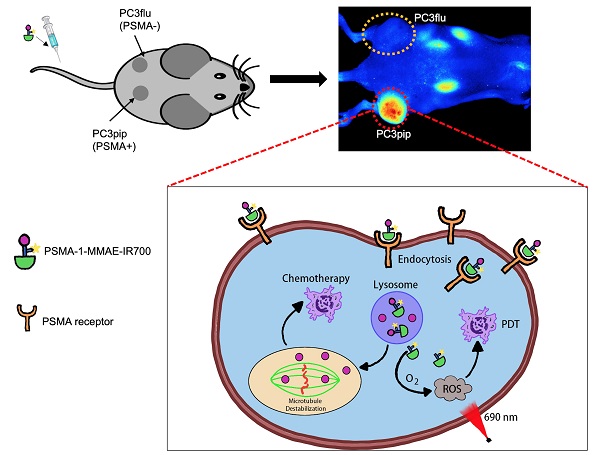
Rationale: Although surgery and radiation therapy in patients with low risk prostate cancer appear appropriate and effective, those with high-risk localized disease almost always become hormone refractory and then rapidly progress. A new treatment strategy is urgently needed for patients with high-risk localized prostate cancer, particularly an approach that combines two drugs with different mechanisms. Combinations of photodynamic therapy (PDT) and chemotherapy have shown synergistic effects in clinical trials, but are limited by off-target toxicity. Prostate specific membrane antigen (PSMA) is a well-established biomarker for prostate cancer. Here we describe the use of a PSMA ligand to selectively and simultaneously deliver a potent microtubule inhibiting agent, monomethyl auristatin E (MMAE), and a PDT agent, IR700, to prostate cancers.
Methods: Using a bifunctional PSMA ligand PSMA-1-Cys-C6-Lys, we created a novel theranostic molecule PSMA-1-MMAE-IR700. The molecule was tested in vitro and in vivo for selectivity and antitumor activity studies.
Results: PSMA-1-MMAE-IR700 showed selective and specific uptake in PSMA-positive PC3pip cells, but not in PSMA-negative PC3flu cells both in vitro and in vivo. In in vitro cytotoxicity studies, when exposed to 690 nm light, PSMA-1-MMAE-IR700 demonstrated a synergistic effect leading to greater cytotoxicity for PC3pip cells when compared to PSMA-1-IR700 with light irradiation or PSMA-1-MMAE-IR700 without light irradiation. In vivo antitumor activity studies further showed that PSMA-1-MMAE-IR700 with light irradiation significantly inhibited PC3pip tumor growth and prolonged survival time as compared to mice receiving an equimolar amount of PSMA-1-IR700 with light irradiation or PSMA-1-IR700-MMAE without light irradiation.
Conclusion: We have synthesized a new multifunctional theranostic molecule that combines imaging, chemotherapy, and PDT for therapy against PSMA-expressing cancer tissues. This work may provide a new treatment option for advanced prostate cancer.
Keywords: prostate cancer, PSMA, MMAE, IR700, multifunctional molecule
 Global reach, higher impact
Global reach, higher impact