Impact Factor
Theranostics 2022; 12(15):6509-6526. doi:10.7150/thno.75431 This issue Cite
Research Paper
MCM6 is a critical transcriptional target of YAP to promote gastric tumorigenesis and serves as a therapeutic target
1. Department of Anatomical and Cellular Pathology, Prince of Wales Hospital, The Chinese University of Hong Kong, Hong Kong SAR, China.
2. Institute of Digestive Disease, State Key Laboratory of Digestive Disease, Li Ka Shing Institute of Health Science, The Chinese University of Hong Kong, Hong Kong SAR, China.
3. State Key Laboratory of Translational Oncology, Sir Y.K. Pao Cancer Centre, The Chinese University of Hong Kong, Hong Kong SAR, China.
4. Department of Anaesthesia and Intensive Care and Peter Hung Pain Research Institute, The Chinese University of Hong Kong, Hong Kong SAR, China.
5. Department of Medicine and Therapeutics, The Chinese University of Hong Kong, Hong Kong SAR, China.
6. Department of Pathology, Nanfang Hospital and Basic Medical College, Southern Medical University, Guangdong Province Key Laboratory of Molecular Tumor Pathology, Guangzhou, China.
7. State Key Laboratory of Genetic Engineering, School of Life Sciences, Zhongshan Hospital, Fudan University, Shanghai, China.
8. Department of Life Science, National Chung Cheng University, Chiayi, Taiwan.
9. School of Biomedical Sciences, The Chinese University of Hong Kong, Hong Kong SAR, China.
10. Department of Clinical Oncology, Hong Kong Cancer Institute, The Chinese University of Hong Kong, Hong Kong SAR, China.
* These authors contributed equally.
Abstract
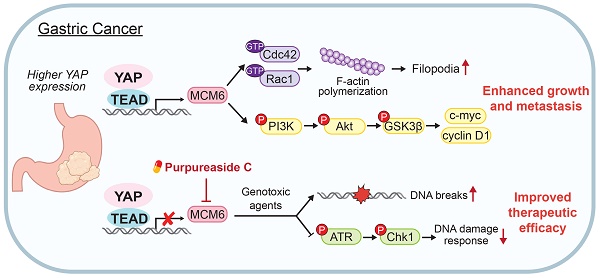
Rationale: Hyperactivation of Hippo-Yes-associated protein (YAP) signaling pathway governs tumorigenesis of gastric cancer (GC). Here we reveal that minichromosome maintenance complex component 6 (MCM6) is a critical transcriptional target of YAP in GC. We aim to investigate the function, mechanism of action, and clinical implication of MCM6 in GC.
Methods: The downstream targets of YAP were screened by RNA sequencing (RNA-seq) and microarray, and further validated by chromatin immunoprecipitation PCR and luciferase reporter assays. The clinical implication of MCM6 was assessed in multiple GC cohorts. Biological function of MCM6 was evaluated in vitro, in patient-derived organoids, and in vivo. RNA-seq was performed to unravel downstream signaling of MCM6. Potential MCM6 inhibitor was identified and the effect of MCM6 inhibition on GC growth was evaluated.
Results: Integrative RNA sequencing and microarray analyses revealed MCM6 as a potential YAP downstream target in GC. The YAP-TEAD complex bound to the promoter of MCM6 to induce its transcription. Increased MCM6 expression was commonly observed in human GC tissues and predicted poor patients survival. MCM6 knockdown suppressed proliferation and migration of GC cells and patient-derived organoids, and attenuated xenograft growth and peritoneal metastasis in mice. Mechanistically, MCM6 activated PI3K/Akt/GSK3β signaling to support YAP-potentiated gastric tumorigenicity and metastasis. Furthermore, MCM6 deficiency sensitized GC cells to chemo- or radiotherapy by causing DNA breaks and blocking ATR/Chk1-mediated DNA damage response (DDR), leading to exacerbated cell death and tumor regression. As there are no available MCM6 inhibitors, we performed high-throughput virtual screening and identified purpureaside C as a novel MCM6 inhibitor. Purpureaside C not only suppressed GC growth but also synergized with 5-fluorouracil to induce cell death.
Conclusions: Hyperactivated YAP in GC induces MCM6 transcription via binding to its promoter. YAP-MCM6 axis facilitates GC progression by inducing PI3K/Akt signaling. Targeting MCM6 suppresses GC growth and sensitizes GC cells to genotoxic agents by modulating ATR/Chk1-dependent DDR, providing a promising strategy for GC treatment.
Keywords: Gastric cancer, YAP, MCM6, PI3K/Akt, therapeutic target.
 Global reach, higher impact
Global reach, higher impact