Impact Factor
Theranostics 2022; 12(15):6762-6778. doi:10.7150/thno.77060 This issue Cite
Research Paper
MnCO3-mineralized polydopamine nanoparticles as an activatable theranostic agent for dual-modality imaging-guided photothermal therapy of cancers
1. Bionanotechnology Research Center, Korea Research Institute of Bioscience and Biotechnology (KRIBB), Daejeon 34141, Republic of Korea
2. Department of Life and Nanopharmaceutical Sciences, Graduate School, Kyung Hee University, Seoul 02447, Republic of Korea
3. Department of Oral Microbiology, School of Dentistry, Kyung Hee University, Seoul 02447, Republic of Korea
4. Department of Maxillofacial Biomedical Engineering, School of Dentistry, Kyung Hee University, Seoul 02447, Republic of Korea
5. Department of Biotechnology, University of Science & Technology (UST), Daejeon 34113, Republic of Korea
Abstract
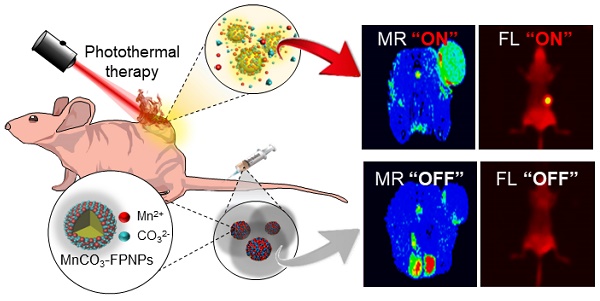
Background: Single imaging modality is still insufficient to evaluate the biological and anatomical structures of tumors with high accuracy and reliability. Generation of non-specific contrast, leading to a low target-to-background signal ratio, results in low imaging resolution and accuracy. Tumor environment-specific activatable multifunctional contrast agents need to maximize the contrast signals, representing a dual imaging-guided photothermal therapy (PTT) at target tumor sites.
Methods: Cellular uptake, cytotoxicity assay, and in vitro photothermal conversion efficiency of MnCO3-mineralized fluorescent polydopamine nanoparticles (MnCO3-FPNPs) were evaluated using 4T1 breast cancer cells. In vivo dual-modality imaging was performed using IVIS imaging and a 4.7 T animal MRI systems after injection into 4T1 tumor-bearing nude mice. The effects of photothermal therapeutic through PTT were measured after irradiation with an 808 nm laser (1.5 W/cm2) for 10 min, measuring the size of the tumors every 2 days.
Results: At physiological pH (7.4), MnCO3-FPNP is efficiently quenched. Conversely, at acidic pH (5.4), the strong fluorescence (FL) is recovered due to the dissociation of Mn2+ from the FPNPs. At pH 7.4, MnCO3-FPNP activity is silenced to enhance water proton relaxation due to unionized MnCO3 maintenance; conversely, at acidic pH (5.4), MnCO3-FPNPs efficiently release Mn2+ ions, thereby resulting in T1-weighted magnetic resonance (MR) contrast enhancement. MnCO3-FPNPs display a promising diagnostic ability for 4T1 breast cancer xenograft models, as well as exhibit a high photothermal conversion efficiency. A successful tumor treatment via their photothermal activity is accomplished within 14 days.
Conclusions: Our studies exhibited unique “OFF-ON” activation abilities in FL/MR dual imaging and PTT functions. This approach suggests that the MnCO3-FPNPs may serve as a useful platform for various mineralization-based multimodal imaging-guided PTT models for many cancer theranostic applications.
Keywords: cancer, fluorescent polydopamine nanoparticles, mineralization, dual-modality imaging, photothermal therapy, theranostics
 Global reach, higher impact
Global reach, higher impact