Impact Factor
Theranostics 2023; 13(2):578-595. doi:10.7150/thno.76614 This issue Cite
Research Paper
Improved antitumor activity against prostate cancer via synergistic targeting of Myc and GFAT-1
1. Center for Pharmacogenetics, Department of Pharmaceutical Sciences, University of Pittsburgh School of Pharmacy, Pittsburgh, PA 15261.
2. Small Molecule Biomarker Core (SMBC), Department of Pharmaceutical Sciences, University of Pittsburgh School of Pharmacy, Pittsburgh, PA 15261.
3. Department of Urology, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261.
#Authors contributed equally to the work.
Abstract
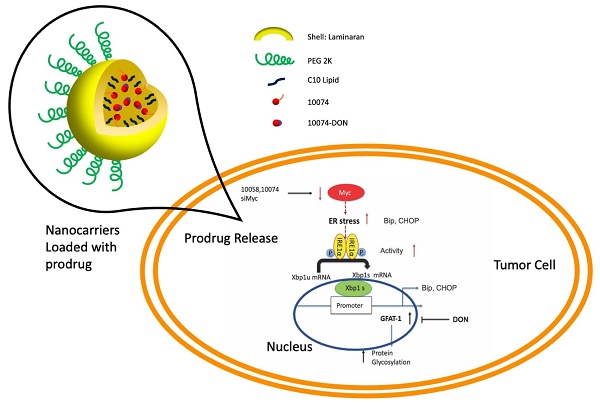
Inhibition of Myc promotes the regression of many types of tumors, including prostate cancer. However, the success of anti-Myc therapy is hampered by the lack of a strategy to effectively deliver the inhibitors to the tumor site and by the feedback mechanisms that cancer cells use to adapt to metabolic reprogramming.
Methods: The effects of Myc inhibitors (10074-G5 or 10058-F4), alone or in combination with 6-diazo-5-oxo-L-norleucine (DON), were evaluated in cultured human or murine prostate cancer cells by cell viability assay, qRT-PCR and Western blot. To facilitate the in vivo therapeutic evaluation, a prodrug conjugate of 10074-G4 and DON (10074-DON) was developed, which could be effectively loaded into a polysaccharide-based nanocarrier (PS).
Results: The treatment with Myc inhibitors led to significant induction of glutamine: fructose-6-phosphate amidotransferase-1 (GFAT1) and enhanced protein glycosylation. Mechanistically, Myc inhibition triggered GFAT1 induction through the IREα-Xbp1s pathway. The combination use of Myc inhibitors and GFAT1 inhibitor DON led to a synergistic effect in inhibiting the proliferation and migration of prostate cancer cells. Enhanced in vivo delivery of 10074-DON via the PS nanocarrier led to a significant inhibition of tumor growth along with an improvement in tumor immune microenvironment in several PCa animal models.
Conclusion: Simultaneous targeting of Myc and GFAT-1 may represent a novel strategy for the treatment of prostate cancer.
Keywords: Myc, glutamine: fructose-6-phosphate amidotransferase-1, protein glycosylation, prostate cancer, therapy
 Global reach, higher impact
Global reach, higher impact