Impact Factor
Theranostics 2023; 13(2):849-866. doi:10.7150/thno.74324 This issue Cite
Research Paper
Kruppel-like factor 2 contributes to blood-spinal cord barrier integrity and functional recovery from spinal cord injury by augmenting autophagic flux
1. Oujiang Laboratory (Zhejiang Lab for Regenerative Medicine, Vision and Brain Health), Department of Orthopaedics Surgery, The Second Affiliated Hospital and Yuying Children's Hospital of Wenzhou Medical University, Wenzhou, 325035, China.
2. Department of Orthopaedics, Affiliated Pingyang Hospital and School of Pharmaceutical Science, Wenzhou Medical University, Wenzhou, 325000, China.
3. Department of Pharmacy, Ningbo Medical Treatment Center Li Huili Hospital, Ningbo, 315040, China.
4. Department of Orthopaedic Surgery, The Third Hospital of Hebei Medical University, Shijiazhuang, 050051 China.
5. Department of Anatomy and Physiology, Shanghai Jiao Tong University School of Medicine, Shanghai, 200025, China.
# These authors contributed equally to this work.
Abstract
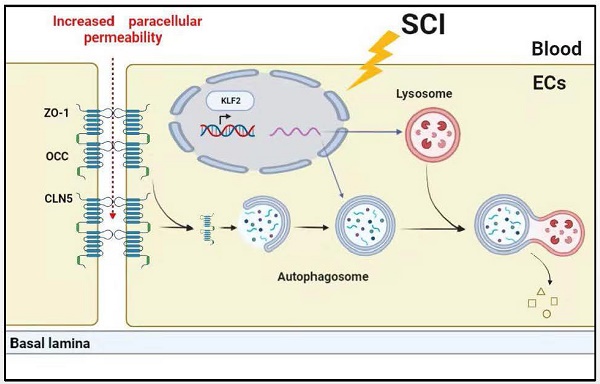
Background: Increasing evidence suggests that acute traumatic spinal cord injury (SCI)-induced defects in autophagy and autophagy-lysosomal pathway (ALP) may contribute to endothelial barrier disruption following injury. Recently, Kruppel-like factor 2 (KLF2) was reported as a key molecular switch on regulating autophagy. Whether KLF2 coordinates endothelial endothelial ALP in SCI is not known.
Methods: Genetic manipulations of KLF2 were performed in bEnd.3 cells and SCI model. Western blot, qRT-PCR, immunofluorescence staining and Lyso-Tracker Red staining, Evans blue dye extravasation, behavioral assessment via Basso mouse scale (BMS), electrophysiology and footprint analysis were performed.
Results: In SCI, autophagy flux disruption in endothelial cells contributes to TJ proteins degradation, leading to blood-spinal cord barrier (BSCB) impairment. Furthermore, the KLF2 level was decreased in SCI, overexpression of which alleviated TJ proteins loss and BSCB damage, which improve motor function recovery in SCI mice, while knockdown of KLF2 displayed the opposite effects. At the molecular level, KLF2 overexpression alleviated the TJ proteins degradation and the endothelial permeability by tuning the ALP dysfunction caused by SCI and oxygen glucose deprivation (OGD).
Conclusions: Endothelial KLF2 as one of the key contributors to SCI-mediated ALP dysfunction and BSCB disruption. KLF2 could be a promising pharmacological target for the management and treatment of SCI.
Keywords: Blood-spinal cord barrier, Tight junction proteins, Spinal cord injury, Kruppel-like factor 2, Autophagy-lysosomal pathway
 Global reach, higher impact
Global reach, higher impact