13.3
Impact Factor
Theranostics 2023; 13(10):3402-3418. doi:10.7150/thno.81700 This issue Cite
Research Paper
Non-viral nitric oxide-based gene therapy improves perfusion and liposomal doxorubicin sonopermeation in neuroblastoma models
1. Department of Biomedical Engineering, University of Texas at Dallas, Richardson, TX, USA.
2. Department of Biological Sciences, University of Texas at Dallas, Richardson, TX, USA.
3. Department of Surgery, University of Chicago Medical School, Chicago, IL, USA.
4. Division of Oncology and Center for Childhood Cancer Research, Children's Hospital of Philadelphia and the Department of Pediatrics and Abramson Cancer Center, University of Pennsylvania, Philadelphia, PA, USA.
Abstract
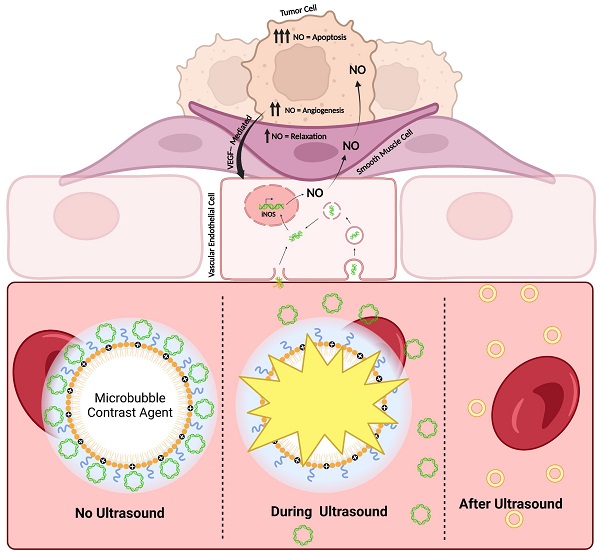
Neuroblastoma (NB) is a pediatric malignancy that accounts for 15% of cancer-related childhood mortality. High-risk NB requires an aggressive chemoradiotherapy regimen that causes significant off-target toxicity. Despite this invasive treatment, many patients either relapse or do not respond adequately. Recent studies suggest that improving tumor perfusion can enhance drug accumulation and distribution within the tumor tissue, potentially augmenting treatment effects without inflicting systemic toxicity. Accordingly, methods that transiently increase tumor perfusion prior to treatment may help combat this disease. Here, we show the use of gene therapy to confer inducible nitric oxide synthase (iNOS) expression solely in the tumor space, using focused ultrasound targeting. NOS catalyzes the reaction that generates nitric oxide (NO), a potent endogenous vasodilator. This study reports the development of a targeted non-viral image-guided platform to deliver iNOS-expressing plasmid DNA (pDNA) to vascular endothelial cells encasing tumor blood vessels. Following transfection, longitudinal quantitative contrast-enhanced ultrasound (qCEUS) imaging revealed an increase in tumor perfusion over 72 h, attributed to elevated intratumoral iNOS expression.
Methods: To construct a gene delivery vector, cationic ultrasound-responsive agents (known as “microbubbles”) were employed to carry pDNA in circulation and transfect tumor vascular endothelial cells in vivo using focused ultrasound (FUS) energy. This was followed by liposomal doxorubicin (L-DOX) treatment. The post-transfection tumor response was monitored longitudinally using qCEUS imaging to determine relative changes in blood volumes and perfusion rates. After therapy, ex vivo analysis of tumors was performed to examine the bioeffects associated with iNOS expression.
Results: By combining FUS therapy with cationic ultrasound contrast agents (UCAs), we achieved selective intratumoral transfection of pDNA encoding the iNOS enzyme. While transitory, the degree of expression was sufficient to induce significant increases in tumoral perfusion, to appreciably enhance the chemotherapeutic payload and to extend survival time in an orthotopic xenograft model.
Conclusion: We have demonstrated the ability of a novel targeted non-viral gene therapy strategy to enhance tumor perfusion and improve L-DOX delivery to NB xenografts. While our results demonstrate that transiently increasing tumor perfusion improves liposome-encapsulated chemotherapeutic uptake and distribution, we expect that our iNOS gene delivery paradigm can also significantly improve radio and immunotherapies by increasing the delivery of radiosensitizers and immunomodulators, potentially improving upon current NB treatment without concomitant adverse effects. Our findings further suggest that qCEUS imaging can effectively monitor changes in tumor perfusion in vivo, allowing the identification of an ideal time-point to administer therapy.
Keywords: gene therapy, inducible nitric oxide synthase (iNOS), sonopermeation, neuroblastoma, and quantitative contrast-enhanced ultrasound (qCEUS)
 Global reach, higher impact
Global reach, higher impact