13.3
Impact Factor
Theranostics 2023; 13(13):4392-4411. doi:10.7150/thno.84427 This issue Cite
Research Paper
DNMT1 mediates the disturbed flow-induced endothelial to mesenchymal transition through disrupting β-alanine and carnosine homeostasis
1. Department of Physiology and Pathophysiology, School of Basic Medical Sciences; Hemorheology Center, School of Basic Medical Sciences, Peking University, Beijing 100191, China.
2. State Key Laboratory of Vascular Homeostasis and Remodeling, Peking University, Beijing 100191, China.
3. National Health Commission Key Laboratory of Cardiovascular Molecular Biology and Regulatory Peptides; Beijing Key Laboratory of Cardiovascular Receptors Research, Peking University, Beijing 100191, China.
Abstract
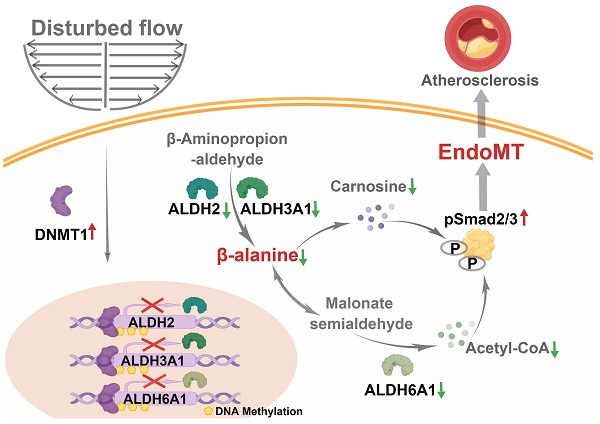
Background: Increasing evidence suggests that hemodynamic disturbed flow induces endothelial dysfunction via a complex biological process so-called endothelial to mesenchymal transition (EndoMT). Recently, DNA methyltransferases (DNMTs) was reported as a key molecular mediator to promote EndoMT. Our understanding of how DNMTs, particularly the maintenance DNMTs, DNMT1, coordinate EndoMT is still lacking.
Methods: A parallel-plate flow apparatus and perfusion devices were used to apply fluid with endothelial protective pulsatile shear (PS, to mimic the laminar flow) or harmful oscillatory shear (OS, to mimic the disturbed flow) to cultured endothelial cells (ECs). Endothelial lineage tracing mice and conditional EC Dnmt1 knockout mice were subjected to a surgery of carotid partial ligation to generate the flow-accelerated atherogenesis models. Western blotting, quantitative RT-PCR, immunofluorescent staining, methylation-specific PCR, chromatin immunoprecipitation, endothelial functional assays, and assessments for neointimal formation and atherosclerosis were performed.
Results: Inhibition of DNMTs with 5-aza-2'-deoxycytidine (5-Aza) suppressed the disturbed flow/OS-induced EndoMT, both in cultured cells and the endothelial lineage tracing mice. 5-Aza also ameliorated the downregulation of aldehyde dehydrogenases (ALDHs) and β-alanine biosynthesis caused by disturbed flow/OS. Knockdown of the ALDH family proteins, ALDH2, ALDH3A1, and ALDH6A1, showed an EndoMT-induction effect as OS. Supplementation of cells with the functional metabolites of β-alanine, carnosine and acetyl-CoA (acetate), reversed EndoMT, likely via inhibiting the phosphorylation of Smad2/3. Endothelial-specific knockout of Dnmt1 protected the vasculature from disturbed flow-induced remodeling and atherosclerosis.
Conclusions: Endothelial DNMT1 acts as one of the key epigenetic factors to mediate the hemodynamically regulated EndoMT at least through repressing the expression of ALDH2, ALDH3A1, and ALDH6A1. Supplementation with carnosine and acetate may have a great potential in the prevention and treatment of atherosclerosis.
Keywords: Endothelial dysfunction, DNA methylation, β-alanine, Carnosine, Hemodynamic disturbed flow
 Global reach, higher impact
Global reach, higher impact