Impact Factor
Theranostics 2015; 5(9):1007-1020. doi:10.7150/thno.11742 This issue Cite
Review
Current Approaches for Improving Intratumoral Accumulation and Distribution of Nanomedicines
1. Department of Molecular Genetics of Intracellular Transport, Institute of Gene Biology, Russian Academy of Sciences, 34/5, Vavilov St., 199334, Moscow, Russia
2. Department of Biophysics, Faculty of Biology, Moscow State University, 1-12, Leninskie Gory, 119991, Moscow, Russia
Abstract
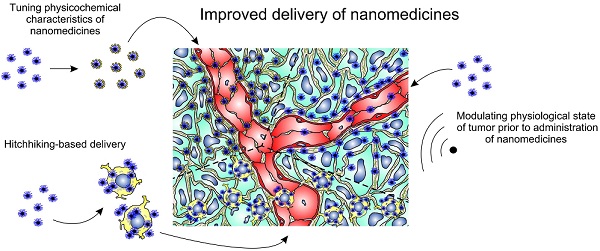
The ability of nanoparticles and macromolecules to passively accumulate in solid tumors and enhance therapeutic effects in comparison with conventional anticancer agents has resulted in the development of various multifunctional nanomedicines including liposomes, polymeric micelles, and magnetic nanoparticles. Further modifications of these nanoparticles have improved their characteristics in terms of tumor selectivity, circulation time in blood, enhanced uptake by cancer cells, and sensitivity to tumor microenvironment. These “smart” systems have enabled highly effective delivery of drugs, genes, shRNA, radioisotopes, and other therapeutic molecules. However, the resulting therapeutically relevant local concentrations of anticancer agents are often insufficient to cause tumor regression and complete elimination. Poor perfusion of inner regions of solid tumors as well as vascular barrier, high interstitial fluid pressure, and dense intercellular matrix are the main intratumoral barriers that impair drug delivery and impede uniform distribution of nanomedicines throughout a tumor. Here we review existing methods and approaches for improving tumoral uptake and distribution of nano-scaled therapeutic particles and macromolecules (i.e. nanomedicines). Briefly, these strategies include tuning physicochemical characteristics of nanomedicines, modulating physiological state of tumors with physical impacts or physiologically active agents, and active delivery of nanomedicines using cellular hitchhiking.
Keywords: nanoparticles, macromolecules, nanomedicines
 Global reach, higher impact
Global reach, higher impact