13.3
Impact Factor
Theranostics 2018; 8(9):2407-2423. doi:10.7150/thno.23632 This issue Cite
Research Paper
Imaging, myeloid precursor immortalization, and genome editing for defining mechanisms of leukocyte recruitment in vivo
1. Institute of Immunology, University of Münster, 48149 Münster, Germany
2. DFG EXC 1003 Cluster of Excellence 'Cells in Motion', University of Münster, Germany
3. European Institute for Molecular Imaging, University of Münster, 48149 Münster, Germany
4. Department of Rheumatology, Radboud University Medical Center, 6525 GA Nijmegen, Netherlands
5. Interdisciplinary Center for Clinical Research, University of Münster, 48149 Münster, Germany
6. Department of Nuclear Medicine, University Hospital Münster, 48149 Münster, Germany
*Both authors contributed equally to this work.
Abstract
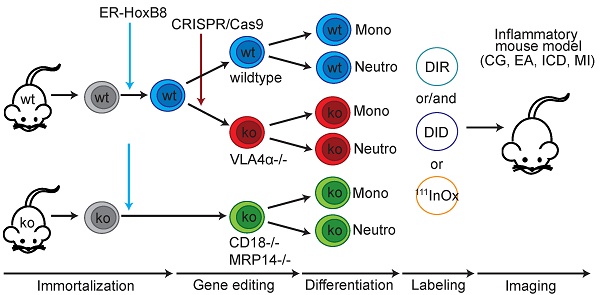
Recruitment of leukocytes from the blood to sites of inflammation poses a promising target for new diagnostic and therapeutic approaches. We aimed to develop a novel method to non-invasively analyze molecular mechanisms of leukocyte migration in pre-clinical models of inflammation in vivo.
Methods: We used the ER-HoxB8 system to transiently immortalize murine myeloid precursors from wildtype and CD18- as well as MRP14-deficient mice. A VLA4α-/- cell line was generated by CRISPR/Cas9-mediated gene editing. We analyzed the migration of wildtype and knockout leukocytes in vivo by optical and nuclear imaging in mice with irritant contact dermatitis, cutaneous granuloma, experimental arthritis and myocardial infarction.
Results: Transient immortalization, gene editing and in vivo imaging can be combined to analyze migratory mechanisms of murine leukocytes, even for gene deletions resulting in lethal phenotypes in mice. We reliably confirmed known migratory defects of leukocytes deficient for the adhesion molecules CD18 or VLA4α. Also, using our new method we identified a new role of the most abundant calcium-binding proteins in phagocytes and major alarmins in many inflammatory diseases, MRP8 and MRP14, for transmigration in vivo.
Conclusion: We provide a combinatorial approach to rapidly characterize molecular mechanisms of leukocyte recruitment in vivo, with the potential to aid in identification of diagnostic and therapeutic targets in inflammatory pathologies.
Keywords: Cell migration, Genetic engineering, molecular imaging, leukocytes, MRP8 and MRP14
 Global reach, higher impact
Global reach, higher impact