Impact Factor
Theranostics 2018; 8(9):2477-2487. doi:10.7150/thno.24173 This issue Cite
Research Paper
Graphene oxide sensitizes cancer cells to chemotherapeutics by inducing early autophagy events, promoting nuclear trafficking and necrosis
1. Department of Chemical Engineering, National Tsing Hua University, Hsinchu, Taiwan 30013
2. Biomedical Technology and Device Research Laboratories, Industrial Technology Research Institute, Hsinchu, Taiwan 31057
3. Institute of Biomedical Engineering, National Chiao Tung University, Hsinchu 30010, Taiwan
4. Department of Biological Science and Technology, National Chiao Tung University, Hsinchu 30010, Taiwan
5. Institute of Molecular Biology, Academia Sinica, Taipei, Taiwan, 11529
6. Department of Biomedical Engineering and Environmental Science, National Tsing Hua University, Hsinchu, Taiwan 30013
7. Frontier Research Center on Fundamental and Applied Sciences of Matters, National Tsing Hua University, Hsinchu, Taiwan 30013
*These two authors contributed equally to this manuscript
Abstract
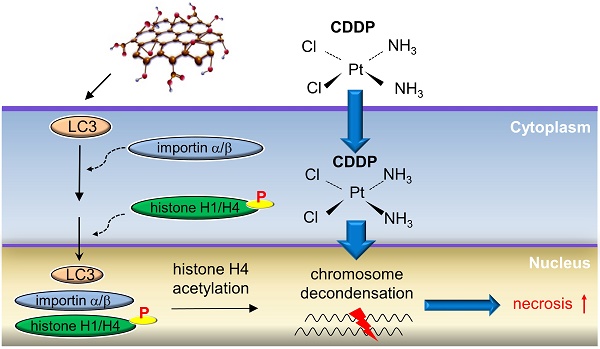
Rationale: Cisplatin (CDDP) is a broad-spectrum anticancer drug but chemoresistance to CDDP impedes its wide use for cancer therapy. Autophagy is an event occurring in the cytoplasm and cytoplasmic LC3 puncta formation is a hallmark of autophagy. Graphene oxide (GO) is a nanomaterial that provokes autophagy in CT26 colon cancer cells and confers antitumor effects. Here we aimed to evaluate whether combined use of GO with CDDP (GO/CDDP) overcomes chemoresistance in different cancer cells and uncover the underlying mechanism.
Methods: We treated different cancer cells with GO/CDDP and evaluated the cytotoxicity, death mechanism, autophagy induction and nuclear entry of CDDP. We further knocked down genes essential for autophagic flux and deciphered which step is critical to nuclear import and cell death. Finally, we performed immunoprecipitation, mass spectrometry and immunofluorescence labeling to evaluate the association of LC3 and CDDP.
Results: We uncovered that combination of GO and CDDP (GO/CDDP) promoted the killing of not only CT26 cells, but also ovarian, cervical and prostate cancer cells. In the highly chemosensitized Skov-3 cells, GO/CDDP significantly enhanced concurrent nuclear import of CDDP and autophagy marker LC3 and elevated cell necrosis, which required autophagy initiation and progression but did not necessitate late autophagy events (e.g., autophagosome completion and autolysosome formation). The GO/CDDP-elicited nuclear trafficking and cell death also required importin α/β, and LC3 also co-migrated with CDDP and histone H1/H4 into the nucleus. In particular, GO/CDDP triggered histone H4 acetylation in the nucleus, which could decondense the chromosome and enable CDDP to more effectively access chromosomal DNA to trigger cell death.
Conclusion: These findings shed light on the mechanisms of GO/CDDP-induced chemosensitization and implicate the potential applications of GO/CDDP to treat multiple cancers.
Keywords: graphene oxide, autophagy, cisplatin, chemoresistance, nuclear import, LC3
 Global reach, higher impact
Global reach, higher impact