Impact Factor
Theranostics 2018; 8(11):3153-3163. doi:10.7150/thno.22080 This issue Cite
Research Paper
A smart bilayered scaffold supporting keratinocytes and muscle cells in micro/nano-scale for urethral reconstruction
1. Department of Urology and Andrology, Shanghai Renji Hospital, Shanghai Jiao Tong University, School of Medicine, Shanghai, China;
2. Department of Urology, Shanghai Jiao Tong University Affiliated Sixth People's Hospital, Shanghai, China;
3. State Key Laboratory for Modification of Chemical Fibers and Polymer Materials, College of Materials Science and Engineering, Donghua University, Shanghai, China;
4. College of Materials and Textile Engineering, JiaXing University, Zhejiang, China.
*Co-first author: These authors contributed equally to the work
Abstract
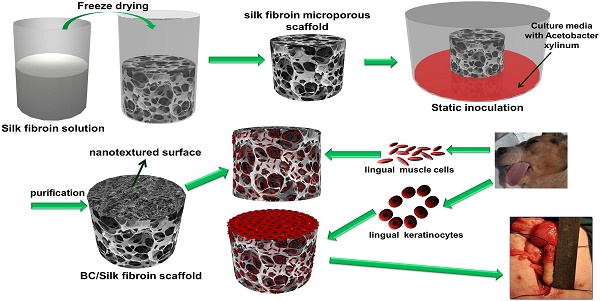
Rationale: In urethral tissue engineering, the currently available reconstructive procedures are insufficient due to a lack of appropriate scaffolds that would support the needs of various cell types. To address this problem, we developed a bilayer scaffold comprising a microporous network of silk fibroin (SF) and a nanoporous bacterial cellulose (BC) scaffold and evaluated its feasibility and potential for long-segment urethral regeneration in a dog model.
Methods: The freeze-drying and self-assembling method was used to fabricate the bilayer scaffold by stationary cultivation G. xylinus using SF scaffold as a template. The surface morphology, porosity and mechanical properties of all prepared SF-BC scaffolds were characterized using Scanning electron microscopy (SEM), microcomputed tomography and universal testing machine. To further investigate the suitability of the bilayer scaffolds for tissue engineering applications, biocompatibility was assessed using an MTT assay. The cell distribution, viability and morphology were evaluated by seeding epithelial cells and muscle cells on the scaffolds, using the 3D laser scanning confocal microscopy, and SEM. The effects of urethral reconstruction with SF-BC bilayer scaffold was evaluated in dog urethral defect models.
Results: Scanning electron microscopy revealed that SF-BC scaffold had a clear bilayer structure. The SF-BC bilayer scaffold is highly porous with a porosity of 85%. The average pore diameter of the porous layer in the bilayer SF-BC composites was 210.2±117.8 μm. Cultures established with lingual keratinocytes and lingual muscle cells confirmed the suitability of the SF-BC structures to support cell adhesion and proliferation. In addition, SEM demonstrated the ability of cells to attach to scaffold surfaces and the biocompatibility of the matrices with cells. At 3 months after implantation, urethra reconstructed with the SF-BC scaffold seeded with keratinocytes and muscle cells displayed superior structure compared to those with only SF-BC scaffold.
Principal Conclusion: These results demonstrate that the bilayer SF-BC scaffold may be a promising biomaterial with good biocompatibility for urethral regeneration and could be used for numerous other types of hollow-organ tissue engineering grafts, including vascular, bladder, ureteral, bowel, and intestinal.
Keywords: bilayer scaffold, bacterial cellulose, silk fibroin, urethral reconstruction, lingual keratinocytes, muscle cells
 Global reach, higher impact
Global reach, higher impact