Impact Factor
Theranostics 2019; 9(6):1741-1751. doi:10.7150/thno.30972 This issue Cite
Research Paper
A Novel TNF-α-Targeting Aptamer for TNF-α-Mediated Acute Lung Injury and Acute Liver Failure
1. Aptamer Core Facility, Institute of Biomedical Sciences, Academia Sinica, Taipei, Taiwan.
2. Institute of Biomedical Sciences, Academia Sinica, Taipei, Taiwan.
3. National Center of Excellence for Clinical Trials and Research Center, Department of Medical Research, National Taiwan University Hospital, Taipei, Taiwan
4. Department of Internal Medicine, National Taiwan University Hospital and College of Medicine, Taipei, Taiwan.
Abstract
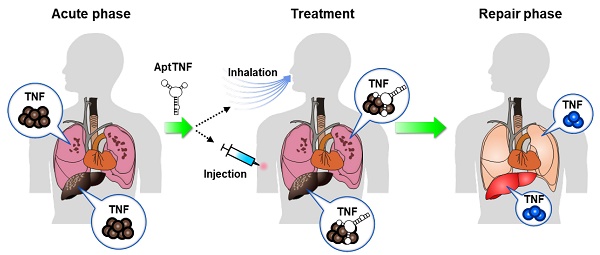
Rationale: The TNF-α pathway plays as a double-edged sword that simultaneously regulates cell apoptosis and proliferation. The dysregulated TNF-α signaling can trigger cytokine storms that lead to profound cell death during the phase of acute tissue injury. On the other hand, an optimal level of TNF-α signaling is required for tissue repair following the acute injury phase. The TNF-α pathway is commonly upregulated in acute lung injury (ALI) and acute liver failure (ALF). Previous studies investigated the feasibility of adopting protein-based TNF-α blockers as disease modifiers in ALI and ALF, but none of these came out with a positive result. One of the potential reasons that resides behind the failure of the trials might be the long half-life of these inhibitors that led to undesired side effects. Developing alternative TNF-α blockers with manageable half-lives remain an unmet need in this regard.
Methods: In the current study, we developed a novel TNF-α-targeting aptamer (aptTNF-α) and its PEG-derivate (aptTNF-α-PEG) with antagonistic functions. We investigated the in vivo antagonistic effects using mouse ALI and ALF models.
Results: Our data showed that aptTNF-α possessed good in vitro binding affinity towards human/mouse TNF-α and successfully targeted TNF-α in vivo. In the mouse ALI model, aptTNF-α/aptTNF-α-PEG treatment attenuated the severity of LPS-induced ALI, as indicated by the improvement of oxygen saturation and lung injury scores, the reduction of protein-rich fluid leakage and neutrophil infiltration in the alveolar spaces, and the suppression of pro-inflammatory cytokines/chemokines expressions in the lung tissues. In the mouse ALF model, we further showed that aptTNF-α/aptTNF-α-PEG treatment not only attenuated the degree of hepatocyte damage upon acute injury but also potentiated early regeneration of the liver tissues.
Conclusion: The results implicated potential roles of aptTNF-α/aptTNF-α-PEG in ALI and ALF. The data also suggested their translational potential as a new category of TNF-α blocking agent.
Keywords: aptamer, acute lung injury, acute liver failure, TNF-α
 Global reach, higher impact
Global reach, higher impact