Impact Factor
Theranostics 2019; 9(7):1825-1836. doi:10.7150/thno.31893 This issue Cite
Research Paper
Gold Clusters Prevent Inflammation-Induced Bone Erosion through Inhibiting the Activation of NF-κB Pathway
1. Department of Chemistry and Chemical Engineering, Beijing University of Technology, Beijing 100124, China
2. CAS Key Laboratory for the Biological Effects of Nanomaterials and Nanosafety, Institute of High Energy Physics, Chinese Academy of Sciences, Beijing 100049, China
Abstract
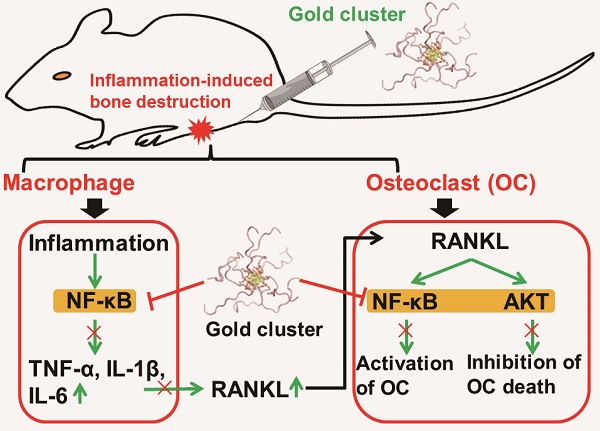
Inflammation-induced bone erosion is a major pathological factor in several chronic inflammatory diseases that often cause severe outcomes, such as rheumatoid arthritis and periodontitis. Plenty of evidences indicated that the inflammatory bone destruction was attributed to an increase in the number of bone-resorbing osteoclasts. However, anti-resorptive therapy alone failed to prevent bone loss in an inflammatory condition. Conventional anti-inflammation treatments are usually intended to suppress inflammation only, but ignore debilitating the subsequent bone destruction. Therefore, inhibition of proinflammatory activation of osteoclastogenesis could be an important strategy for the development of drugs aimed at preventing inflammatory bone destruction.
Methods: In this study, we synthesized a peptide coated gold cluster to evaluate its effects on inflammatory osteoclastogenesis in vitro and inflammation-induced bone destruction in vivo. The in vitro anti-inflammation and anti-osteoclastogenesis effects of the cluster were evaluated in LPS-stimulated and receptor activator of nuclear factor κB ligand (RANKL) stimulated macrophages, respectively. The LPS-induced expression of crucial pro-inflammation cytokines and RANKL-induced osteoclastogenesis as well as the activation of NF-κB pathway in both situations were detected. The inflammation-induced RANKL expression and subsequent inflammatory bone destruction in vivo were determined in collagen-immunized mice.
Results: The gold cluster strongly suppresses RANKL-induced osteoclast formation via inhibiting the activation of NF-κB pathway in vitro. Moreover, treatment with the clusters at a dose of 5 mg Au/kg.bw significantly reduces the severity of inflammation-induced bone and cartilage destruction in vivo without any significant toxicity effects.
Conclusion: Therefore, the gold clusters may offer a novel potent therapeutic stratagem for inhibiting chronic inflammation associated bone destruction.
Keywords: gold cluster, osteoclastogenesis, inflammatory bone destruction, NF-κB pathway
 Global reach, higher impact
Global reach, higher impact