Impact Factor
Theranostics 2019; 9(7):1837-1850. doi:10.7150/thno.27051 This issue Cite
Research Paper
An Optogenetic Controllable T Cell System for Hepatocellular Carcinoma Immunotherapy
1. The United Innovation of Mengchao Hepatobiliary Technology Key Laboratory of Fujian Province, Mengchao Hepatobiliary Hospital of Fujian Medical University, Fuzhou 350025, P. R. China
2. Liver Disease Center, The First Affiliated Hospital of Fujian Medical University, Fuzhou 350005, P. R. China
3. The Liver Center of Fujian Province, Fujian Medical University, Fuzhou 350025, P. R. China
4. College of Life Science, Fujian Agriculture and Forestry University, Fuzhou, 350002, P.R. China
5. Department of Comparative, Fuzhou General Hospital, Xiamen University Medical College, 156 Road Xi'erhuanbei, Fuzhou 350025, Fujian, P.R. China
*These authors contributed equally to this work.
Abstract
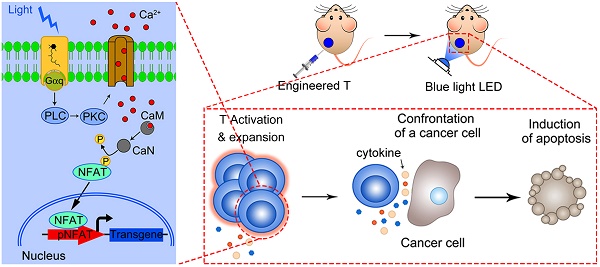
Rationale: T-cell based immunotherapy increasingly shows broad application prospects in cancer treatment, but its performance in solid tumors is far from our expectation, partly due to the re-inhibition of infiltrated T cells by immunosuppressive tumor microenvironment. Here we presented an artificial synthetic optogenetic circuit to control the immune responses of engineered T cells on demand for promoting and enhancing the therapeutic efficiency of cancer immunotherapy.
Methods: We designed and synthesized blue-light inducible artificial immune signaling circuit and transgene expression system. The blue light triggered transgene expression was investigated by luciferase activity assay, qPCR and ELISA. The in vitro cytotoxicity and proliferation assays were carried out on engineered T cells. The in vivo anti-tumor activity of engineered T cells was investigated on xenograft model of human hepatocellular carcinoma.
Results: Blue light stimulation could spatiotemporally control gene expression of specific cytokines (IL2, IL15, and TNF-α) in both engineered 293T cells and human primary T cells. This optogenetic engineering strategy significantly enhanced the expansion ability and cytolytic activity of primary T cells upon light irradiation, and the light activated T cells showed high-efficiency of elimination against xenograft of hepatocellular carcinoma cells.
Conclusions: The current study represented an engineered remotely control T cell system for solid tumor treatment, and provided a potential strategy to partially overcome the intrinsic shortages of current immune cell therapy.
Keywords: optogenetics, engineered T cells, hepatocellular carcinoma, immune cell therapy
 Global reach, higher impact
Global reach, higher impact