13.3
Impact Factor
Theranostics 2019; 9(7):2100-2114. doi:10.7150/thno.29538 This issue Cite
Research Paper
Gastric cancer cells escape metabolic stress via the DLC3/MACC1 axis
1. Department of Oncology, Nanfang Hospital, Southern Medical University, Guangzhou, China;
2. Department of Cardiology, Nanfang Hospital, Southern Medical University, Guangzhou, China.
3. Karolinska Institute, Department of Medicine-Solna, Clinical Pharmacology Group, Karolinska University Hospital-Solna, Stockholm, Sweden.
*These authors contributed equally to the study.
Abstract
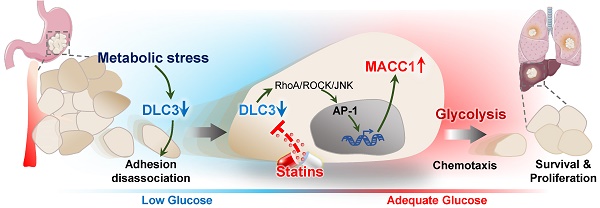
Metabolic stress usually occurs in rapidly growing gastric cancer (GC) when the energy demand exceeds the supply. Interestingly, cancer cells can somehow escape this stress. Some small Rho GTPases regulating cell migration can be activated by metabolic stress. DLC3 is a RhoA-specific GTPase-activating protein of unclear function in cancer. We hypothesized that it participated in metabolic stress escape.
Methods: Metabolic stress in GC cells was induced by glucose deprivation, and DLC3 expression was detected. Based on the prognostic value, cell viability, motility and glycolysis were detected in DLC3 differently expressed GC cells in vitro and in vivo. DLC3 downstream targets were screened and verified. Chemotactic ability was evaluated to study DLC3 and its downstream signaling on metabolic stress escape. In addition, therapeutic strategies targeting DLC3 were explored.
Results: DLC3 expression was lowered by metabolic stress in GC cells. DLC3 downregulation indicated poor cancer prognosis, and silencing DLC3 promoted GC cell proliferation and invasion. MACC1, an oncogene promoting GC growth and metastasis, was proved to be the downstream target of DLC3. Low DLC3 expression and high MACC1 expression indicated high recurrence rate after GC resection. DLC3 transcriptionally inhibited MACC1 expression via RhoA/JNK/AP-1 signaling, and subsequently suppressed GC cell glycolysis and survival under metabolic stress. The DLC3/MACC1 axis modulated the chemotaxis of GC cells from energy deficient area to glucose abundant area. Finally, lovastatin was found to be a promising therapeutic drug targeting the DLC3/MACC1 axis.
Conclusions: The DLC3/MACC1 axis modulates GC glycolysis and chemotaxis to escape glucose deprivation. Lovastatin may inhibit GC by targeting the DLC3/MACC1 axis.
Keywords: DLC3, MACC1, metastasis, chemotaxis, lovastatin
 Global reach, higher impact
Global reach, higher impact