13.3
Impact Factor
Theranostics 2019; 9(9):2662-2677. doi:10.7150/thno.32060 This issue Cite
Research Paper
A self-assembled, modular nucleic acid-based nanoscaffold for multivalent theranostic medicine
1. Interdisciplinary Nanoscience Center (iNANO), Aarhus University, Gustav Wieds Vej 14, 8000 Aarhus C, Denmark
2. Biomolecular Nanoscale Engineering Center, Department of Physics, Chemistry and Pharmacy, University of Southern Denmark, Campusvej 55, 5230 Odense M, Denmark
Abstract
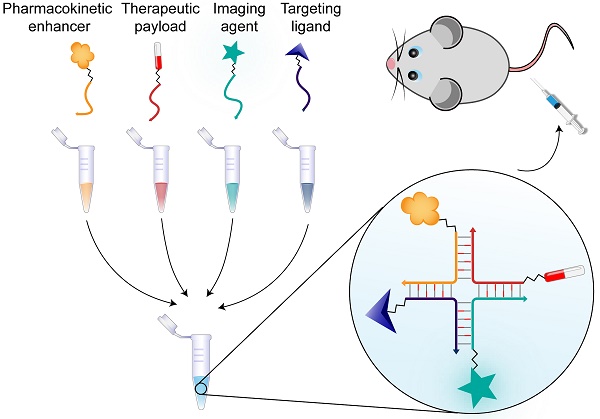
Rationale: Within the field of personalized medicine there is an increasing focus on designing flexible, multifunctional drug delivery systems that combine high efficacy with minimal side effects, by tailoring treatment to the individual.
Methods: We synthesized a chemically stabilized ~4 nm nucleic acid nanoscaffold, and characterized its assembly, stability and functional properties in vitro and in vivo. We tested its flexibility towards multifunctionalization by conjugating various biomolecules to the four modules of the system. The pharmacokinetics, targeting capability and bioimaging properties of the structure were investigated in mice. The role of avidity in targeted liver cell internalization was investigated by flow cytometry, confocal microscopy and in vivo by fluorescent scanning of the blood and organs of the animals.
Results: We have developed a nanoscaffold that rapidly and with high efficiency can self-assemble four chemically conjugated functionalities into a stable, in vivo-applicable system with complete control of stoichiometry and site specificity. The circulation time of the nanoscaffold could be tuned by functionalization with various numbers of polyethylene glycol polymers or with albumin-binding fatty acids. Highly effective hepatocyte-specific internalization was achieved with increasing valencies of tri-antennary galactosamine (triGalNAc) in vitro and in vivo.
Conclusion: With its facile functionalization, stoichiometric control, small size and high serum- and thermostability, the nanoscaffold presented here constitutes a novel and flexible platform technology for theranostics.
Keywords: LNA, drug delivery, bioconjugation, nanomedicine, personalized medicine
 Global reach, higher impact
Global reach, higher impact