13.3
Impact Factor
Theranostics 2019; 9(12):3425-3442. doi:10.7150/thno.33178 This issue Cite
Research Paper
Knockdown of NEAT1 induces tolerogenic phenotype in dendritic cells by inhibiting activation of NLRP3 inflammasome
1. Department of Cardiology, The Second Affiliated Hospital of Harbin Medical University, Harbin, China
2. The Key Laboratory of Myocardial Ischemia, Harbin Medical University, Ministry of Education, Harbin, China
3. Department of Thoracic Surgery, Harbin Medical University Cancer Hospital, Harbin, China
*These authors contributed equally to this work.
Abstract
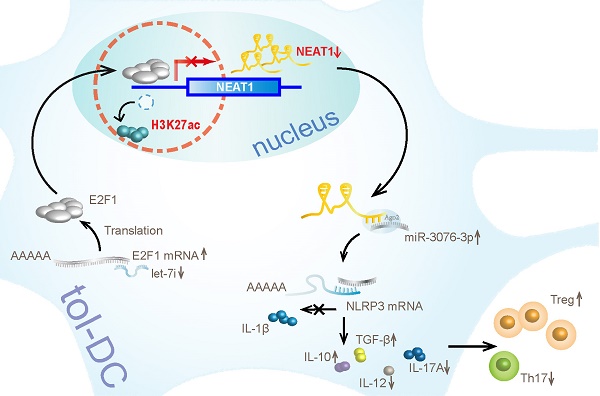
Rationale: Tolerogenic dendritic cells (tol-DCs) play essential roles in immune-related diseases and induce immune tolerance by shaping T-cell responses. Accumulating evidence suggests that long noncoding RNAs (lncRNAs) play important regulatory roles in the immune system. However, the potential roles and underlying mechanisms of lncRNAs in tol-DCs remain unclear.
Methods: RNA in-situ hybridization, histochemistry, and qRT-PCR were performed to determine the distribution and expression of NEAT1 in DCs. Flow cytometry was used to analyze the tolerogenic function of DCs. Small sequencing, followed by bioinformatic analysis, was performed to determine the target genes of NEAT1. The mechanism of NEAT1 was explored using a luciferase reporter, chromatin immunoprecipitation assays, and Immunofluorescence. In-vivo experiments were used to investigate the induction of immune tolerance via NEAT1-knockdown DCs.
Results: Our results show that lncRNA NEAT1 can induce tolerogenic phenotype in DCs. Mechanistically, small RNA-seq analysis revealed that NEAT1 knockdown preferentially affected the expression of miR-3076-3p. Furthermore, NEAT1 used the NLRP3 inflammasome as a molecular decoy for miR-3076-3p, thus facilitating the expression of tolerogenic phenotype in DCs. Moreover, the transcription factor E2F1 acted as a repressor of NEAT1 transcription via activity of H3K27ac. Our results also indicate that NEAT1 knockdown in DCs can induce immune tolerance in models of experimental autoimmune myocarditis and heart transplantation.
Conclusions: Taken together, our study shows the mechanism used by NEAT1 in inducing tol-DCs and highlights the therapeutic potential of targeting NEAT1 for the treatment of immune-related diseases.
Keywords: tolerogenic dendritic cells, immune tolerance, long non-coding RNA, NEAT1
 Global reach, higher impact
Global reach, higher impact