Impact Factor
Theranostics 2019; 9(12):3659-3673. doi:10.7150/thno.32126 This issue Cite
Research Paper
RIP3 promotes colitis-associated colorectal cancer by controlling tumor cell proliferation and CXCL1-induced immune suppression
1. National Translational Science Center for Molecular Medicine, 710032 Xi'an, China.
2. Department of Cell Biology, School of Basic Medicine, The Fourth Military Medical University, 710032 Xi'an, China.
* These authors contributed equally to this work.
Abstract
Rationale: Necroptosis is a programmed form of non-apoptotic cell death that requires receptor-interacting protein 3 (RIP3). RIP3 has been shown to be relevant in multiple tumor types and has differential impact on tumor progression. We investigated whether RIP3 is involved in the progression of colitis-associated cancer (CAC) in mice.
Methods: Tissues from colorectal cancer patients were examined for RIP3 expression. CAC was induced using azoxymethane (AOM) injection followed by dextran sodium sulfate (DSS) treatment in RIP3-deficient or wild-type mice. Colon tissues were collected and analyzed by Western blotting and gene expression profile analyses. Immune cell infiltration and CXCL1 expression were examined by flow cytometry and Real-time PCR, respectively.
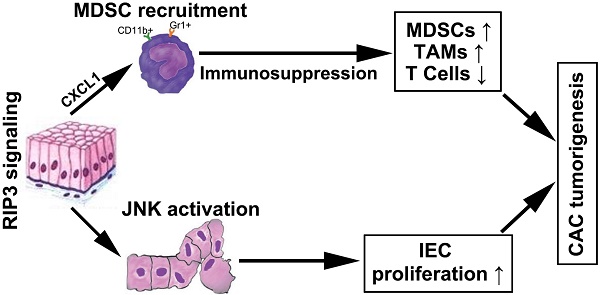
Results: RIP3 expression was upregulated in mouse CAC and human colon cancer. RIP3-deficient mice showed significantly attenuated colitis-associated tumorigenesis. Bone marrow transplantation experiments suggested that RIP3's function in hematopoietic cells primarily contributes to the phenotype. RIP3 supported epithelial proliferation and tumor growth via JNK signaling but had no effect on apoptosis. RIP3 deletion increased T cell accumulation and reduced infiltration by immunosuppressive subsets of myeloid cells during acute colitis and CAC. The immune-suppressive tumor microenvironment was dependent on RIP3-induced expression of the chemokine attractant CXCL1, and administration of recombinant CXCL1 during CAC restored tumorigenesis in Rip3-/- mice.
Conclusion: Our results reveal an unexpected function of RIP3 in enhancing the proliferation of premalignant intestinal epithelial cells (IECs) and promoting myeloid cell-induced adaptive immune suppression. These two distinct mechanisms of RIP3-induced JNK and CXCL1 signalling contribute to CAC progression.
Keywords: colorectal cancer, IBD, ulcerative colitis, necroptosis, RIP3.
 Global reach, higher impact
Global reach, higher impact