Impact Factor
Theranostics 2019; 9(14):4115-4129. doi:10.7150/thno.35131 This issue Cite
Research Paper
Combination Therapy of TGF-β Blockade and Commensal-derived Probiotics Provides Enhanced Antitumor Immune Response and Tumor Suppression
1. National Engineering Research Center for Nanomedicine, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan, 430074, China
2. Key Laboratory of Molecular Biophysics of Ministry of Education, Center for Human Genome Research, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan, 430074, China
3. Department of Biotechnology, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan, 430074, China
4. Department of Microbiology, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA 19104, USA
5. Hepatic Surgery Center, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430030, China
#These authors contributed equally to this work.
Received 2019-3-22; Accepted 2019-5-7; Published 2019-5-31
Abstract
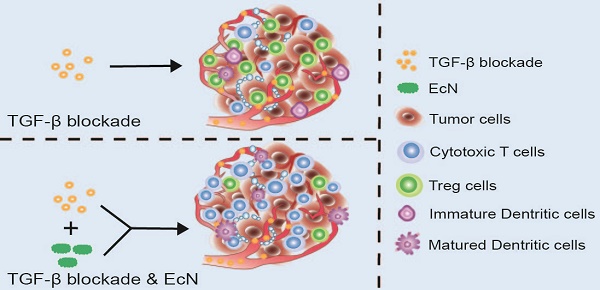
Galunisertib (Gal) is a transforming growth factor (TGF-β) blockade which is being investigated as a potential tumor immunotherapy candidate drug in clinical trials. However, primary or acquired resistance is often found in the recruited cancer patients, which limits its clinical application. Tumor immune microenvironment can be regulated by intestinal microbiota, leading to different therapeutic outcomes. It is hypothesized that manipulation of cancer patients' intestinal microbiome in the early stage of therapy may be a promising strategy to improve the therapeutic efficacy of Gal.
Methods: 4T1 and H22 subcutaneous tumor bearing mice were used to evaluate the therapeutic effect. Escherichia coli strain Nissle 1917 (EcN), a widely used probiotic bacteria, was orally delivered to the tumor bearing mice daily along with Gal treatment. Antitumor effect of the combination therapy was evaluated by tumor volume, histological staining of tumor tissues. Furthermore, flow cytometry was performed to analyze the alteration of immune microenvironment in tumor bed after treatment. The suppressing effect of the combination therapy on tumor invasiveness and metastasis was evaluated in both mice and zebrafish xenografts models. Fecal sample 16S rRNA gene sequencing was conducted to analyze changes of intestinal microbial diversity. The effect of intestinal microbiota on tumor suppression after receiving EcN was further tested by fecal transplant.
Results: The therapeutic outcomes in tumor growth inhibition and metastasis suppression of Gal were significantly potentiated by EcN, resulting from the strengthened antitumor immunity. EcN was able to relieve the immunosuppressive tumor microenvironment, which was evidenced by enhanced tumor-specific effector T cells infiltration and dendritic cells activation. Intestinal microbiota was modulated by EcN, illustrated by a shift of gut microbiome toward certain beneficial bacteria.
Conclusion: These results suggested that Gal combined with EcN might be a novel therapeutic approach with great potential of clinical implications for cancer prevention or treatment.
Keywords: TGF-β blockade, immunotherapy, gut microbiota, cancer treatment, tumor immune microenvironment
Introduction
Cancer immunotherapy has aroused general concern and becomes the most promising therapeutic strategy for hematological neoplasms and solid malignancies, mainly including antibody therapy, cellular immunotherapy, cytokine therapy and so forth [1, 2]. The immune surveillance is stimulated by immunotherapy in cancer patients to make neoantigens be recognized and eradicated better [3, 4]. Antibody-based therapeutics, especially immune checkpoint inhibitors (ICIs) that target the PD-1/PD-L1 axis or CTLA-4, have shown unprecedented clinical efficacy [5, 6]. Transforming growth factor β (TGF-β) is a multifunctional immunosuppressive cytokine that plays a crucial role in tumorigenesis, facilitates the escape of tumors from immune surveillance and promotes tumor invasion and metastasis, rendering it a possible target for cancer immunotherapy [7]. Gal (also known as LY2157299 monohydrate), an orally delivered small molecule antagonist of TGF-β receptor I, has already proved its efficacy on tumor control in pre-clinical studies [8]. However, there are still a considerable proportion of cancers which cannot be successfully treated or controlled by ICIs or TGF-β blockade therapy [9]. In fact, only a minority of patients with nonselective solid tumors can benefit from such immunotherapies but still with a risk of recurrence [10, 11]. In this case, it is urgent and necessary to establish strategies to improve the clinical response and therapeutic efficacy of current cancer immunotherapy.
Drugs for cytokine immunotherapy, such as TGF-β blockade, often do not target tumor cells directly but boost immune system in a more general way [7]. Although the blockade could somehow impact TGF-β responsive cells such as fibroblastic, endothelial, and immune cells in tumor microenvironment (TME), it may have quite limited effects on tumor cells expressing altered or non-functional TGF-β signal [12]. It is speculated that the discrepancy of clinical outcomes of TGF-β blockade among patients partly results from the wide variations of TME in different individuals or some host-related influences [7, 8]. Hence, researchers are attempting to combine the blockade with other standard treatment regimens to achieve better therapeutic effects. For instance, Gal is administrated along with nivolumab to participants with advanced refractory solid tumors, refractory non-small cell lung cancers (NSCLCs) or hepatocellular carcinoma (HCC) to enhance immune response against tumor cells in the ongoing clinical trials [13].
Increasing evidences have demonstrated that gut microbiota can be utilized to potentiate the anti-tumor efficacy of cancer immunotherapies [14, 15]. Commensal bacteria has been revealed to promote the occurrence and development of malignant tumor by influencing both local and systemic immune systems [16, 17]. The dysbiosis of gut microbiome was proved to affect the therapeutic efficacy of PD-1 blockade in tumor bearing mice and cancer patients, while oral supplementation with specific strains could improve the antitumor immune response [18]. In addition, researchers also found that the antitumor effects of CTLA-4 blockade depended on the gut microbiota, especially on the Bacteroides species, indicating innovative strategies to ameliorate such immunotherapies [19]. Therefore, it is indicated that manipulation of gut microbiota might be an effective way to improve the clinical outcomes of cancer immunotherapy. Up to now, the role of intestinal microbiota in animals or cancer patients receiving TGF-β blockade still remains unknown. We hypothesize that intestinal microbiota may serve as a potential approach to restore the therapeutic efficiency of TGF-β blockade.
Escherichia coli strain Nissle 1917 (EcN), a most investigated versatile commensal bacteria with strain specific properties and proven efficacy, has been successfully used to treat chronic inflammatory disorders and strengthen the gut barrier functions in clinical applications [20, 21]. It is the only probiotic recommended by European Crohn´s and Colitis Organization guidelines as effective alternative treatment to alleviate ulcerative colitis from exacerbations and registered as a drug in some European countries [22]. Furthermore, EcN has been shown to be equivalent to mesalazine which brought sustained relief in ulcerative colitis patients without carrying any pathogenic factors [22]. Multiple mechanisms have been demonstrated to support the above-mentioned beneficial effects, including inhibition of pathogen adhesion and invasion to epithelial cells, modulation of immune responses and maintaining integrity of the mucosal barrier [21, 22].
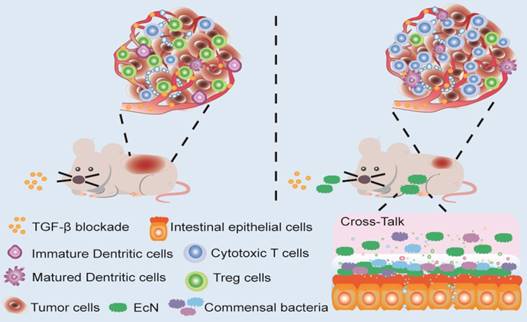
Here in this study, we try to explore whether EcN could be used to enhance the therapeutic effects of TGF-β blockade. Antitumor effects were evaluated in tumor bearing mice administered with Gal in combination with EcN. The underlying mechanism including alteration of TME was further elucidated. Besides, we aim to determine whether the antitumor immune efficiency induced by EcN is achieved through regulating commensal microbiota. Our finding provided the first basis that EcN could improve the therapeutic effect of TGF-β blockade through modulating intestinal commensal microbiota and TME, thus opening up new avenues for cancer immunotherapy (Scheme 1).
Schematic illustration of EcN facilitating TGF-β blockade efficacy in tumor bearing mice. Combination treatment with TGF-β blockade and EcN could markedly trigger tumor suppression in subcutaneous tumor bearing mice, which was stronger than TGF-β blockade treated alone. The enhanced efficiency of the combinational therapy was accompanied by promotion of effector T cells infiltration and relief of immune suppression in tumor bed, which may be the result of cross-talk between intestinal commensal bacteria and host immune system.
Methods
Animals and cell culture
Balb/c mice were purchased from Hubei Province Center for Disease Control and Prevention (Wuhan, China) at 6-8 weeks (18-20 g) of age. All mice were housed in a specific pathogen-free environment at a constant temperature (22 ± 3℃), with a 12 h light/dark cycle and fed adaptively for 1 week after arrival. During the experiments, mice had free access to water and were fed the maintenance diet (China National Cereals, Oils and Foodstuffs Corporation, China). All animals received humane care and experimental protocols were carried out in accordance with the Guide for the Care and Use of Laboratory Animals of Huazhong University of Science and Technology, as approved by the Animal Care Committee of Hubei Province.
Murine hepatic carcinoma cell line H22 and breast carcinoma cell line 4T1 (acquired from Type Culture Collection of Chinese Academy of Sciences, Shanghai, China) were cultured in RPMI-1640 medium (Gibco, Thermo Fisher Scientific Inc., USA.), supplemented with 10% bovine calf serum and 1% antibiotics (100 units/mL penicillin and 100 μg/mL streptomycin) at 37 °C in a 5% CO2 incubator.
Bacterial culture and drug intervention
EcN (non-pathogenic probiotic isolate (serotype O6: K5: H1), Mutaflor) was kindly provided by Jun Zhu, University of Pennsylvania. A nonpathogenic human commensal intestinal bacteria Escherichia coli strain MG1655 (K-12 reference strain), was used as a control for EcN. Strains were grown and maintained in lysogeny broth under aerobic condition as previously described [23]. TGF-β blockade (Gal), also named as LY2157299, was purchased from Shengda Pharmaceutical Co., Ltd. (Shenzhen, China).
Subcutaneous tumor model in mice and dosage regimen
In HCC model, mice were inoculated subcutaneously with injections of 2×106 H22 tumor cells into their right flanks. In murine breast cancer model, mice were challenged subcutaneously with 1×106 4T1 tumor cells instead. Mice in each model were randomly divided into 4 groups (n = 6). For the EcN group, each mouse was daily treated with 1×109 CFU (suspended in 200 µL of saline) by oral gavage on the day of tumor inoculation for 22 days in HCC model and 27 days in breast cancer model, respectively. For the Gal group, mice were orally administrated with Gal at a dose of 45 mg/kg for 7 consecutive days after tumor volume reached approximately 80~100 mm3. For the combination therapy group, mice were treated with Gal along with EcN as described above, respectively. For the control group, 200 µL of saline was daily administrated to mice by oral gavage. Tumor volume and body weight were recorded every 3 days. Two-dimensional tumor volume was measured with a digital vernier calliper. The tumor volume was calculated using the formula: tumor volume = length × width2 × 0.5. When the tumor volume reached about 2000 mm3, mice were sacrificed according to the guidelines for animal care. Tumors were isolated and weighed. In addition, fundus vein blood samples were collected for further study. Fecal samples were collected, snap frozen in sterile cryovial immediately and stored at -80 °C for subsequent analysis.
Antibiotic treatment and fecal transplantation
Mice in the antibiotic treatment group received a cocktail of broad-spectrum antibiotics including vancomycin (0.1 g/L), neomycin (0.2 g/L), ampicillin (0.2 g/L), and metronidazole (0.2 g/L) in their drinking water for 1 week [24]. Fresh drinking water containing antibiotics was replaced every other day. Subsequently, fecal samples collected from EcN-respond tumor-bearing mice were administrated by oral gavage to each antibiotics-treated mouse every 2 days until the tumor volume reached about 2000 mm3. To prepare the stool samples, 1 g of the mixed fresh fecal pellet was suspended in 5 mL of saline. The suspensions were vortexed and then centrifuged at 3000 rpm for 2 min. An aliquot of 200 μL supernatant was collected for transplantation to each mouse.
Histological staining and evaluation
Partial tumor tissues were fixed in 4% paraformaldehyde, sectioned and stained with H&E. The rest of tumor tissues were frozen and prepared to be stained with Ki67 and TUNEL according to the manufacturer's instructions. Cell nuclei were stained by DAPI. Images of the stained sections were acquired by fluorescence microscopy (Olympus, IX51).
Flow cytometric analysis
For analysis of tumor infiltrating lymphocytes, mice tumors were isolated, digested in collagenase I and filtered into single cell suspensions [25]. Afterwards, the suspensions were stained with FITC-labeled anti-mouse CD3 antibody (clone 145-2C11, cat. 100203, 1:50 dilution), PerCP/Cy5.5-labeled anti-mouse CD4 antibody (clone GK1.5, cat. 100434, 1:80 dilution) and APC-labeled anti-mouse CD8a antibody (clone 53-6.7, cat. 100712, 1:80 dilution). For intracellular cytokine staining, lymphocytes prepared from tumor mass were restimulated using phorbol 12-myristate 13-acetate (PMA; 50 ng/mL) and ionomycin (1 mg/mL; Sigma-Aldrich) in the presence of Brefeldin A (1 mg/mL; eBioscience) for 4 h. Subsequently, the simulated lymphocytes were stained with FITC-labeled anti-mouse CD3 antibody and APC-labeled anti-mouse CD8a antibody as described above. After surface staining, cells were treated with Fix/Perm solution and restained with PE-labeled anti-mouse IFN-γ antibody (clone XMG1.2, cat. 505807, 1:80 dilution). For preliminary phenotypic analysis of Tregs, tumor infiltrating lymphocyte were stained with PerCP/Cy5.5-labeled anti-mouse CD4 antibody and APC-labeled anti-mouse CD25 antibody (clone PC 61, cat. 102011, 1:80 dilution). For dendritic cell analysis, tumor infiltrating lymphocytes were stained with APC-labeled anti-mouse CD11c antibody (clone N418, cat. 117310, 1:80 dilution) and PE-labeled anti-mouse I-A/I-E antibody (clone M5/114.15.2, cat. 107607, 1:80 dilution). All antibodies were purchased from BioLegend (San Diego, USA), and flow cytometric analysis was performed by using a CytoFlex flow cytometry system (Beckman Coulter, USA).
Microbial DNA Extraction and Sequencing
Mice stool were frozen at -80 °C immediately after collection. Total genomic DNA from around 100 mg of stool was extracted by QIAamp DNA Stool Mini Kit (QIAGEN, Valencia, CA) according to the manufacturer's guide. The V3-V4 region of the bacterial 16S ribosomal RNA (rRNA) genes was amplified by PCR with universal primer sequences (338F, ACTCCTACGGGAGGCAGCAG; 806R, GGACTACHVGGGTWTCTAAT) and FastPfu Polymerase. Amplicons were then purified by gel extraction (AxyPrep DNA GelExtraction Kit, Axygen Biosciences, USA) and were quantified using QuantiFluor-ST (Promega, USA). The purified amplicons were pooled in equimolar concentrations, and paired-end sequencing was performed using an Illumina MiSeq platform (Illumina, San Diego, USA).
Zebrafish Xenografts Implantation and Treatment
Zebrafish Tg (kdrl:mcherry, neovascularization possessed in red fluorescence) was purchased from the Institute of Hydrobiology, Chinese Zebrafish Resource Center of Chinese Academy of Sciences. Zebrafish embryos were kept in a 28.5 °C incubator and all the protocols were handled according to the guidelines from the Laboratory Animal Training Association.
Within 24 hours post-fertilization (hpf), embryos of zebrafish were incubated with standard embryo media. During the experiment, the embryos were not fed, but were maintained by yolk-derived nutrients. At 48 hpf, zebrafish embryos were stripped and anesthetized with 0.04 mg/mL of tricaine (MS-222, Sigma). Anesthetized embryos were transferred onto a modified agarose gel for microinjection. For the EcN treatment group, EcN was grown overnight in Luria Broth (LB) shaking at 37 °C. Prior to inoculation, bacteria suspensions were centrifuged at 8,000 g for 2 min and subsequently washed twice with embryo medium cultures. Immediately after the embryo was acquired, it was exposed to the inoculum of 1×106 CFU/mL EcN until the end of the experiments. For the TGF-β blockade treatment group, 4T1 tumor cells were pre-incubated with 0.3μg/mL Gal for 48 h. For the control group and EcN treated group, 4T1 tumor cells for microinjection were treated with PBS instead of Gal. Afterwards, about 400 of Dio-labeled tumor cells were injected into the perivitelline space of each zebrafish at 48 hpf. After injection, xenografted zebrafish were transferred to 24-well plates and maintained in 30 °C incubator until the end of experiments. Fluorescence microscope (Olympus, IX71) was used to evaluate the micrometastasis behavior in vivo. Tumor cells in zebrafish often tend to metastasize from head to tail, so the caudal vessels of xenografts with micrometastasis were focused for detailed imaging under confocal microscope (Olympus, FV1000) at 4th day post injection (4 dpi). Image J was used to calculate the percentage of xenografts with micrometastasis.
Statistical Analysis
Statistical analysis was programmed by Student's t-test or one-way analysis of variance (ANOVA). Results are presented as the mean ± SD. Differences were considered significant at *P < 0.05, **P < 0.01, respectively.
Results
Therapeutic efficiency of combination administration of TGF-β blockade and EcN in mice
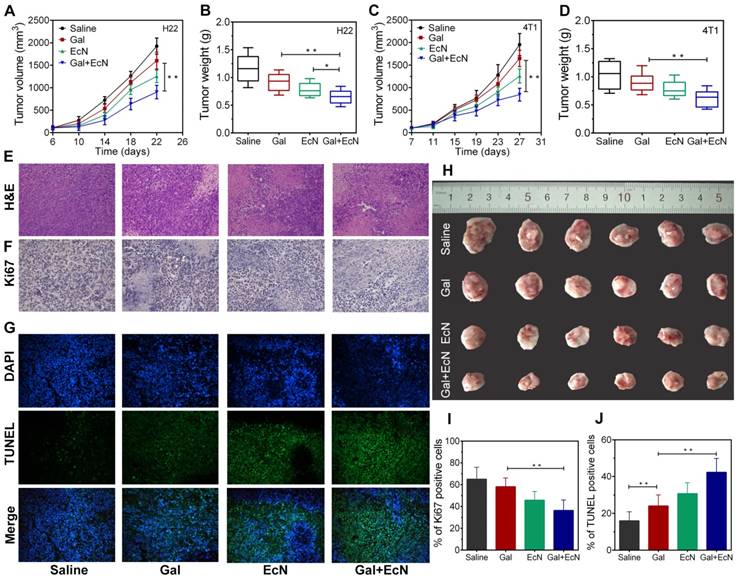
To investigate whether TGF-β blockade combining EcN can trigger better tumor control, the antitumor effect was initially evaluated after EcN administration by detecting the tumor volume change both in HCC and breast carcinoma subcutaneous tumor model. As shown in Figure 1A-B, pretreatment with EcN can slow down the tumor promotion in H22 tumor bearing mice compared to the controls (P < 0.05) and appeared to be more effective than Gal treated alone. Meanwhile, combined treatment of Gal and EcN could markedly delay tumor growth compared to single treatment with Gal. Besides, compared to control group without any treatment, more extensive necrosis within the tumor was illustrated by H&E staining in combinational therapy group (Figure 1E). In addition, the immunofluorescence of Ki67 in Figure 1F reflects the relatively less proliferation in the combination treatment. Consistently, TUNEL assay displayed a more serious apoptosis in tumor tissues harvested from tumor bearing mice treated with Gal and EcN as compared to the Gal treated or EcN treated alone, with an average apoptosis index of 43.5 %, compared to the control of 18.8 % (Figure 1G). Simultaneously, such similar expected outcomes from combined treatment were also observed in 4T1 murine breast cancer (Figure 1C-D). Besides, no acute body weight drop was observed from the above treatments throughout experiment (Figure S1). Together, these data demonstrated that the combination of TGF-β blockade and EcN may generate a better antitumor efficiency.
EcN enhanced the anti-tumor effects of TGF-β blockade in tumor-bearing mice. (A) Tumor growth in H22 hepatic carcinoma cell bearing mice (n = 6). (B) Tumor weight of H22 hepatic carcinoma cell bearing mice at the end of the experiment. (C) Tumor growth in 4T1 breast carcinoma cell bearing mice (n = 6). (D) Tumor weight of 4T1 breast carcinoma cell bearing mice at the end of the experiment. (E to G) Representative histological analysis of tumor specimens stained by H&E, Ki67, DAPI (blue) and TUNEL (green) at 200×, data are representative of two independent experiments (n = 3). (H) Representative images of tumor tissues at the end of the experiment. (I) Semi-quantification of proliferation index of the tumor tissue in the Ki67 staining assay. (J) Semi-quantification of apoptosis index of the tumor tissue in the TUNEL staining assay. All data are shown as mean ± SD. (* P < 0.05, ** P < 0.01)
Combination therapy of TGF-β blockade and EcN induced the activation of spontaneous antitumor-specific immune responses
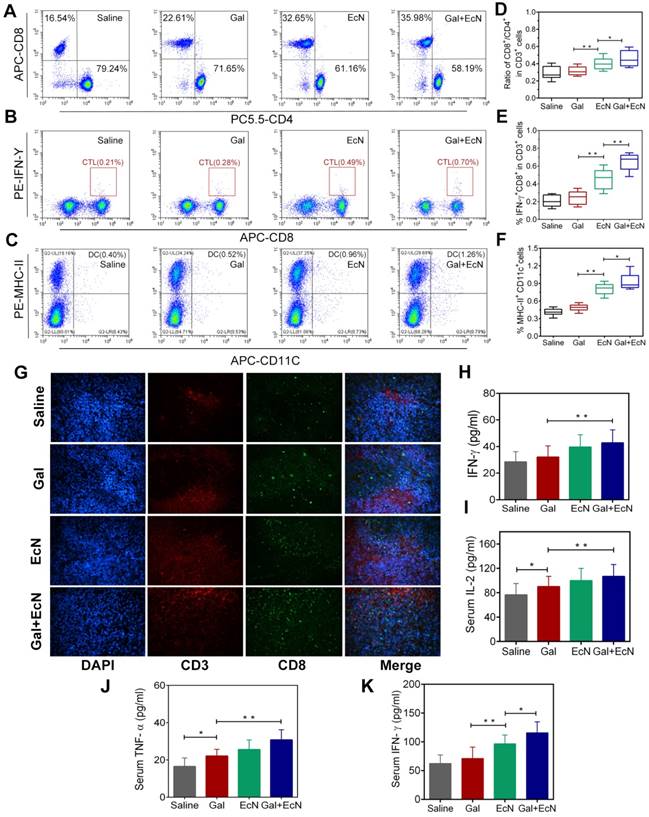
Harnessing the host immune system contributes to a satisfactory cancer treatment outcome. Commensal bacterial provide immunomodulatory signals during the immune system development and immune cell activation, which are crucial to shaping systemic immune responses and homeostasis. In this regard, we assumed that the combination antitumor effect might be immune-mediated. To verify this hypothesis, tumor-infiltrating lymphocytes were harvested and evaluated between different groups. Administration with EcN alone or in combination with TGF-β blockade could increase the percentage of CD8+/CD3+ T cells, while decrease the percentage of CD4+/CD3+ T cells in tumor tissues (Figure 2A and 2D). It was indicated that EcN treatment could enhance tumor-specific T cell responses and intratumoral CD8+ T cell accumulation after systemic treatment. Meanwhile, the combination treatment could also effectively stimulate the proliferation and activation of cytotoxic T lymphocytes (CTLs) by recruiting a higher proportion of IFN-γ+CD8+ cells in CD3+ T cells in tumor-draining lymph nodes, thus leading to a favorable immunotherapy outcome (Figure 2B and 2E). Besides, we also found larger number of mature dendritic cells (DCs) in tumor-draining lymph nodes in combination therapy (Figure 2C and 2F). More powerful DCs maturation and infiltration presumably resulted in stronger antitumor activity after combinational application of TGF-β blockade and EcN. Moreover, representative images of immunofluorescent staining from tumor tissues illustrate that EcN combined with TGF-β blockade therapy significantly promoted the antitumor-specific lymphocyte infiltration into TME (Figure 2G). Importantly, sustained administration of TGF-β blockade and EcN resulted in a significant improvement in subcutaneous transplantation tumor immune microenvironment, demonstrated by the significantly elevated levels of IFN-γ in tumor tissues as well as TNF-α and IL-2 levels in the serum (Figure 2H-K).
EcN enhanced the effects of TGF-β blockade in activation of antitumor-specific immune responses in tumor-bearing mice. (A) Representative flow cytometry analysis of CD8+ cells and CD4+ cells in CD3+ T cells in tumor tissues (n = 4-6). (B) Representative flow cytometry analysis of IFN-γ+ cells and CD8+ in CD3+ T cells in tumor-draining lymph nodes. (C) Representative flow cytometry analysis of MHC-II+ cells in CD11c+ cells in tumor infiltration lymph nodes (n = 4-6). (D) Ratio of CD8+ /CD4+ cells in CD 3+ T cells in tumor tissues. (E) Ratio of IFN-γ+ CD8+ T cells in CD3+ T cells in tumor-draining lymph nodes. (F) Ratio of MHC-II+ cells in CD11c+ cells in tumor infiltration lymph nodes. (G) Representative immunofluorescence analysis of CD3+ and CD8+ T cells from tumor sections at 200× . ELISA measurement of IFN-γ (H) in tumor tissue homogenate from treated mice (n = 6). ELISA measurement of IL-2 (I), TNF-α (J) and IFN-γ (K) in serum from treated mice (n = 6). All data are shown as mean ± SD. (* P < 0.05, ** P < 0.01)
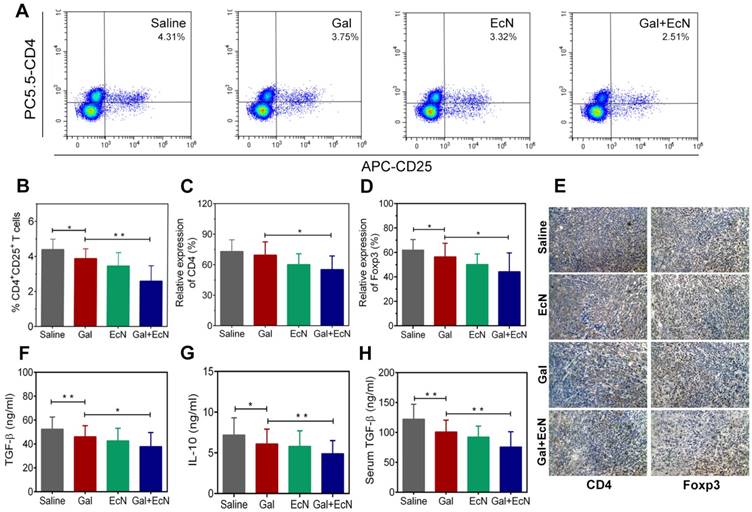
Antitumor effect of TGF-β blockade and EcN was correlated with the remodulation of immunosuppressive tumor microenvironment
Tumor development is profoundly influenced by its local immune microenvironment and an immunosuppressive microenvironment can favor tumor cell growth and metastasis. To investigate whether the combination therapy could improve the immune surveillance escape and thus contribute to the antitumor effect, we analyzed the proportion of regulatory T cells (Tregs) in tumor tissues. Flow cytometry analysis revealed a significant decrease in the ratio of CD4+CD25+ T cells in tumor tissues for combination treatment compared to TGF-β blockade or EcN-treated alone (Figure 3A-B). Consistently, immunohistochemical staining of CD4+ and Foxp3+ Tregs in tumor tissues confirmed that there were fewer Foxp3+ Tregs in tumors from combination treated mice compared to the tumor bearing controls (Figure 3C-E). Importantly, administration of TGF-β blockade with EcN resulted in a significant improvement in the subcutaneous TME, demonstrated by a significant reduction in the level of immuno-inhibitory cytokines such as TGF-β and IL-10 both in the serum and tumor tissue homogenate (Figure 3F-H). These results suggest that the combined treatment could transfer the tumor milieu from immuno-inhibitory to immune-stimulatory state, which may improve the tumor immune microenvironment, thus restoring the efficiency of TGF-β blockade.
EcN enhanced the effects of TGF-β blockade in the remodulation of immunosuppressive tumor microenvironment in tumor-bearing mice. (A) Representative flow cytometry analysis of CD4+CD25+ regulatory T cells in tumor tissues (n = 4-6). (B) Analysis of CD4+CD25+ regulatory T cells in tumor tissues. Semi-quantification of CD4+ (C) and Foxp3+ Tregs (D) in tumor sections. (E) Representative images of immunohistochemical staining of CD4+ and Foxp3+ Tregs in tumor specimens at 200× (n = 3). Measurement of TGF-β (F) and IL-10 (G) in tumor tissue homogenates by ELISA (n = 6). (H) Measurement of TGF-β in mice serum by ELISA (n = 6). All data are shown as mean ± SD. (* P < 0.05, ** P < 0.01)
TGF-β blockade with EcN influenced tumor invasion and metastasis behavior in mice and zebrafish xenografts model
Metastasis requires cancer cells to escape from primary tumor, survive in the circulation, and grow at distant sites, which is closely related to tumor immune microenvironment. Herein, anti-tumor metastasis efficiency of combination therapy was evaluated. As shown in Figure 4A, due to the solid tumor nodules, the lung weight (Figure 4D) of mice in TGF-β blockade group was heavier than that of combined treatment group. Meanwhile, the numbers of metastasis nodules on the surface of lungs in combined treatment group were decreased (Figure 4E), suggesting that combination administration of TGF-β blockade with EcN could remarkably and effectively reverse the promoted metastasis. H&E staining further verified that physiological structure of the lung in combination therapy tended to be normal with fewer micrometastatic lesions observed (Figure 4B). We further evaluated the microvessel density with angiogenesis marker (CD31+) by immunohistochemical staining. Results revealed a significant decrease of relative number of vascular sprouts between combination therapy and TGF-β blockade or EcN treated alone (Figure 4C and F), indicating that TGF-β blockade with EcN treatment might limit tumor metastasis by weakening angiogenesis.
EcN facilitated anti-invasion and anti-metastasis of TGF-β blockade in tumor-bearing mice and cancer cell xenografted zebrafish. (A) Representative images of the lungs from mice bearing subcutaneous tumor (n = 4-6). The white arrows pointed the metastatic nodules. (B) Histological analysis of the lung sections by H&E staining at 100× (n = 3). (C) Expression of CD31, an angiogenesis marker, in representative tumor sections by immunostaining at 200× (n = 3). (D) Wet weight of the lungs from mice bearing subcutaneous tumor (n = 4-6). (E) Numbers of superficial metastatic nodules in the lung tissues from mice bearing subcutaneous tumor (n = 4-6). (F) Analysis of CD31+ cells in tumor tissues from different treated tumor-bearing mice. (G) Quantification of the percentage of xenografts with micrometastasis. (H) Fluorescence images (100×) of zebrafish tail at 4 dpi. (I) Schematic representation of xenotransplantation in zebrafish, 4T1 tumor cell were labeled with Dio and injected into the perivitelline space of zebrafish embryos. All data are shown as mean ± SD. (* P < 0.05, ** P < 0.01)
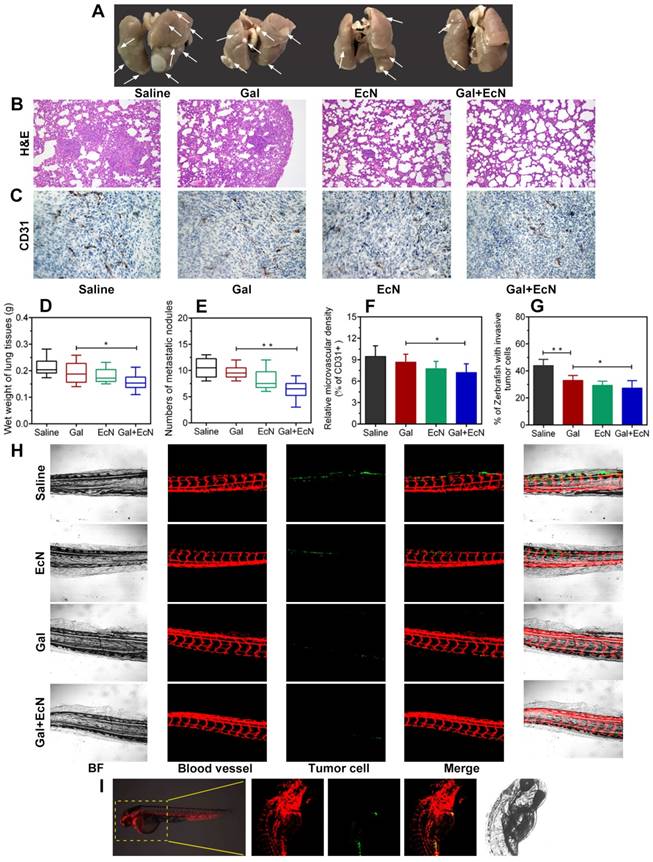
Transgenic zebrafish have increasingly been developed into a powerful model for organogenesis and cancer biology, providing a unique tool to track tumor cell behavior in vivo and monitor tumor-induced invasion in response to drug treatments in real time during cancer progression (Figure 4I). Thus, we evaluated 4T1 cell metastasis ability in zebrafish xenografts model among different groups (Figure 4H). Our results revealed that tumor cell tended to spread and colonize at the caudal vessels in saline-treated control group which showed higher metastasis capacity. However, we found that TGF-β blockade with EcN therapy could induce lower percent of zebrafish showing invading tumor cells (Figure 4G) and suppresses micrometastasis (Figure 4H) compared to TGF-β blockade treated alone. Besides, in order to further validate whether EcN could actually be taken up by zebrafish in our study, tumor cells were marked with Dir, EcN was labeled with green fluorescence protein (GFP) and added directly to the embryo medium, and then we repeated the previous microinjection experiment (Figure S2). The results showed that EcN could get into the intestinal tract of zebrafish where it played a potential role by interacting with the intestinal immune system.
Antitumor efficiency of EcN was strain specific and commensal bacterial mediated
In order to explore the possible mechanism of how EcN exerts immune-mediated antitumor efficiency, nonpathogenic intestinal Escherichia coli strain MG1655 and heat-inactivated EcN were performed in subcutaneous HCC model. Nevertheless, similar tumor control effects were not observed when tumor bearing mice were exposed to the MG1655 (Figure 5A), which indicated that the tumor control capacity of EcN was probably strain-specific. Meanwhile, it is interesting to note that heat inactivation of EcN before oral administration could dramatically diminish the therapeutic effect on tumor growth (Figure 5A). To confirm whether the therapeutic effect is dominated by using EcN alone or interacting with other commensal bacteria, depletion of conventional gut microbiota was implemented in mice with antibiotic cocktail. Notably, we found that EcN compromised its therapeutic effect in mice which were basically depleted of gut microbiota (Figure 5B).
Antitumor efficiency of EcN in tumor-bearing mice is strain specific and partly mediated by commensal bacteria. (A) Tumor growth in mice treated with heat-inactivated EcN or nonpathogenic intestinal Escherichia coli strain MG1655. (B) Tumor growth in mice pre-treated with antibiotics. (C) Tumor growth in mice co-housed with EcN treated mice. (D) Tumor growth in mice receiving fecal transplantation from EcN treated mice. (E) Tumor growth in mice treated with EcN-focA-. All data are shown as mean ± SD. (n = 6, * P < 0.05, ** P < 0.01)
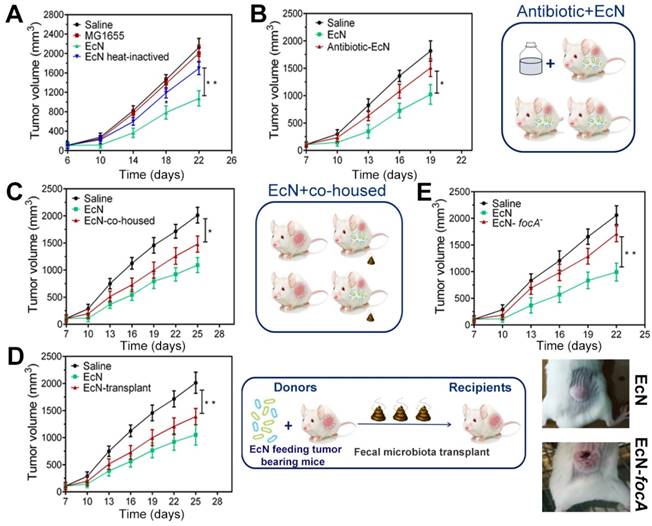
To further address whether this difference could be mediated by commensal microbiota, we cohoused EcN-respond mice and saline-treated tumor bearing mice from tumor implantation to the end of the experiment. We found that after cohoused with EcN-respond mice, the tumor bearing controls appeared to acquire the EcN-respond mice phenotype, resulting in triggering its tumor rejection, which suggested that tumor bearing controls may be colonized by commensal microbes that dominantly facilitated antitumor immunity (Figure 5C). Furthermore, to directly test the role of commensal bacteria in regulating EcN antitumor immunity, feces from EcN-respond mice were collected and transplanted to the tumor bearing controls by oral gavage daily (Figure 5D). Our study indicated that transfer of EcN-respond tumor bearing mice fecal material, but not saline, into recipients was sufficient to elicit effective tumor suppression, which supported a microbe-derived effect in cancer immunotherapy.
All through the way, EcN is a robust colonizer of the human intestine. focA, encoding the major F1C fimbrial subunit, was known to be responsible for biofilm formation of EcN and epithelial cell adherence. Here, focA was knockout by λ-Red recombination method which was performed with modification as previously described (Figure S3) [23]. We found that the focA mutant strain showed a remarkable deficiency in tumor control compared to the unmodified strains (Figure 5E). In addition, we did not find the similar downward trend in tumor-bearing mice by oral gavage with culture supernatants, suggesting that these effects may not be related to their direct metabolites (Figure S4).
Oral administration of EcN mediated changes of gut microbiota profile in tumor bearing mice
Changes in the structure of the microflora were visualized by partial least squares discriminant analysis (PLS-DA) in Figure 6A, which revealed that the overall bacteria community from EcN-respond tumor bearing mice gradually deviated from the tumor bearing controls over time. Moreover, the richness of gut microbiota, measured by numbers of observed operational taxonomic units (OTUs), was increased in the EcN-respond mice compared to the controls (Figure 6B).
Changes of microbial diversities in EcN-respond mice compared with tumor bearing controls. (A) Partial least squares discriminant (PLS-DA) analysis of fecal samples between EcN-respond mice and tumor bearing controls. (B) Numbers of observed OUTs in EcN-respond mice and tumor bearing controls. Microbiome alterations at the genus level between EcN-respond mice and tumor bearing controls: relative abundant of EcN-respond enriched genera Bacteroides (C), Akkermansia (D), Alistipes (E) and Oscillibacter (F). Microbiome alterations at the species level between EcN-respond mice and tumor bearing controls: Alistipes shahii (G), Akkermansia muciniphila (H), Bacteroides thetaiotaomicron (I) and Clostridium_sp (J). (K) Spearman correlation heat map analysis of the relationships between relative abundance of EcN-respond associated bacteria and the percentage of tumor infiltrating CTLs, IFN-γ level in the serum, TGF-β or IL-10 level in tumor tissue. All data are shown as mean ± SD. (n = 6, * P < 0.05, ** P < 0.01)
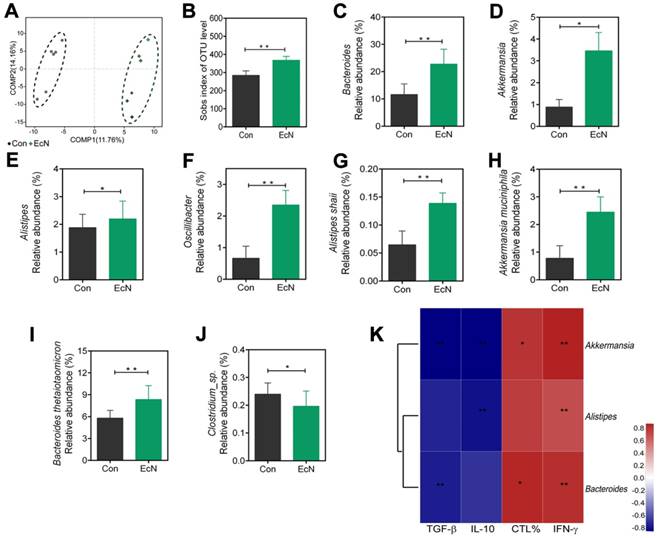
To investigate the specific changes of microbiota among EcN-respond mice, we assessed the changes in the relative abundance of taxa between EcN-respond mice and tumor bearing controls. At the genus level, we observed bacterial taxa that displayed different abundance between EcN-respond mice and tumor bearing controls (Figure S5). Bacteroides, as the most abundant genus in both groups, was dramatically increased in EcN-respond mice compared with tumor bearing controls (Figure 6C). Of the remaining taxa, three genera including Allstipes (Figure 6E), Akkermansia (Figure 6D) and Oscillibacter (Figure 6F) were also enriched in EcN-respond mice. Furthermore, at the species level, we also found that Alistipes shahii (Figure 6G), Akkermansia muciniphila (Figure 6H), Bacteroides thetaiotaomicron (Figure 6I), Bacteroides acidifaciens and Lactobacillus johnsonii (Figure S5) were increased in EcN-respond mice compared with tumor bearing controls. Conversely, Clostridium_sp. (Figure 6J) was decreased in EcN-respond mice. Moreover, spearman correlation analysis was used to investigate the correlation between the relative abundance of EcN-respond associated species and the ratio of tumor infiltrating CTLs, IFN-γ level in serum, TGF-β and IL-10 levels in tumor tissue. We observed a significant positive correlation between Akkermansia muciniphila and IFN-γ level in serum while a negative correlation with immunosuppressive cytokines TGF-β and IL-10, suggesting a positive association with antitumor T cell responses (Figure 6K). We thus hypothesized that members of this species could represent a major component of the beneficial antitumor immune effects observed in EcN-respond mice.
Discussion
Tumor cells often secrete a variety of immunosuppressive cytokines, including TGF-β and IL-10, to outgrow and evade host immune surveillance, giving the rational for tumor immunotherapy which aims to stimulate our own immune system to fight against cancer [4]. TGF-β pathway inhibitors, as an emerging strategy for cancer therapy, may contribute to reverse this microenvironment-induced immune suppression by inhibiting Tregs, restoring natural killer (NK) cells and T cell-mediated cytotoxicity [7]. However, the antitumor activity of TGF-β blockade alone is limited [26]. In addition, it appears that only selected tumor cells could respond to TGF-β blockade treatment in standard or immune-deficient xenografts [27, 28]. In the present study, we also found that oral delivery of Gal alone showed mild effect on tumor burden or tumor immune microenvironment in tumor bearing mice. It is partly due to the lack of tumor targeting of TGF-β blockade by systemic administration and also the fact that the inhibitory function is largely dependent on patients' own immune conditions [28, 29].
Tumorigenesis, a complex and multi-dynamic process, including initiation, progression, and metastasis, is closely related to TME [30]. TME is created by the tumor and dominated by tumor-induced interactions. Tumor cells can influence the local microenvironment by releasing extracellular signals, promoting angiogenesis and inducing peripheral immune tolerance, while immune cells in the microenvironment can affect the growth and evolution of cancer cells [31]. Immune cells present in TME include T lymphocytes, DCs, macrophages as well as other innate immune cells. In most cases, although various immune cells are recruited to the tumor site, their antitumor functions are often in dormancy [31]. Instead, they are assigned to promote tumor growth, leading to the exhaustion of CTLs, enrichment in Tregs and myeloid derived suppressor cells (MDSCs) as well as other immunosuppressive cytokines [32]. Among TME, tumor-infiltrating lymphocytes (TILs) usually represented as the major component of immune infiltrates within tumors. TILs obtained from human tumor tissues showed inhibited proliferation in response to tumor antigens, compromised signaling through T cell receptors and decreased ability to mediate cytotoxicity of tumor targets [33]. Thus, there is a need to develop novel therapeutic strategy which target to remodeling the TME into a microenvironment that is beneficial for acute response and effective antitumor activity.
By influencing local immune homeostasis and systematically immune regulatory with second immune organs, specific commensals may have synergetic functions with cancer therapy, such as chemotherapy, radiotherapy, immunotherapy and so forth. For immunotherapy, resident microorganisms are known to directly or indirectly impact host-targeted therapy and facilitate drug efficacy [34, 35]. For example, cyclophosphamide could lead to the translocation of Lactobacillus johnsonii and Lactobacillus murinus into mesenteric lymph nodes, where they were conducive to stimulate the Th1 and Th17 immune response in tumor-bearing mice model. However, in germ-free mice or antibiotic treated mice, it failed to generate this response and proved resistant to the drug [36]. Notably, single dose with Bifidobacterium on 7th and 14th days after tumor implantation could protect against tumor initiation as effective as anti PD-L1 therapy, shedding light on the non-negligible role of specific commensal bacteria in tumor control [37].
EcN has been proven to be beneficial for intestinal immune homeostasis, resulting in efforts to repair the intestinal dysfunction and restore the intestinal barrier function [21]. The presence and composition of specific bacterial species not only influence the local mucosal immune homeostasis, but also affect the physiological functions and the response to therapy in distant organs, systemically [38]. Our work found that oral administration of EcN alone could inhibit tumor growth, which is probably due to the increased tumor-specific effector T cell infiltration and improved tumor immunosuppression. Besides, this study highlights the therapeutic potential of administered TGF-β blockade in combination with EcN in cancer treatment and their global influences in distant tumor beds. The combination treatment in our work seemed to restore the function of tumor infiltrating CTLs to destroy cancer cells, elevate levels of IFN-γ, IL-1β, TNF-α in the serum and increase the proportion of tumor infiltrating CD3+CD8+ IFN-γ+ CTLs. Meanwhile, DCs are generally considered as an important mediator in priming tumor antigen presentation process, resulting in the upregulation of lymphocytes recruitment, effective cytokines production and promotion T cells proliferation [4]. In this work, we also found an increased percentage of activated DCs from tumor-draining lymph nodes in combination therapy. Taken together, our data suggested that EcN-derived signals might induce DCs activation, which in turn supports improved effector function of tumor-specific CTLs.
Intriguingly, oral administration of MG1655 had poor effect on tumor control, suggesting that the antitumor immunity effect should depend on the specific strain. Besides, we found that EcN lose its therapeutic effect of tumor control in antibiotic treated mice, indicating that the therapeutical effect of using EcN alone is limited. Instead, it needs to cooperate with other specific commensal bacterial. The intestinal homeostasis might be the basis of bacterial-immune synergy for response to TGF-β blockade therapy. EcN was thought to be a good colonizer and biofilm former in human intestine [39]. Mounting evidences have shown that bacterial biofilm formation plays an important role in intestinal colonization, forming cross-talk with epithelial cells and other resident microbiota [23]. Notably, in our current study, tumor suppression deficiency was found in focA mutant strain, indicating that EcN should only be colonized in the intestine before it could have the capacity to interact with other commensal bacteria, and thus exert synergistic therapeutic tumor immunomodulatory activity.
To identify the specific bacteria associated with improved antitumor immune responses, we analyzed the differences between EcN-respond mice and tumor bearing controls through the 16S ribosomal RNA MiSeq Illumina platform. The gut microbiome of EcN-respond mice reflected enriched species richness as well as a significant shift in the overall microbial diversity compared to tumor bearing control. Recent studies revealed the important role of Alistipes shahii in the gut as a modulator positively associated with immuno-stimulatory cytokines TNF-α production in TILs and thus suppress tumor growth [40]. Our findings showed an increase of Alistipes shahii in EcN-respond mice, suggesting that the beneficial effect of combined treatment with TGF-β blockade and EcN in delaying tumor growth were related to increase TNF-α production and thus to awaken CTLs in tumor beds. Bacteria within the Bacteroides in general, or specifically, Bacteroides thetaiotaomicron and Bacteroides fragilis were identified as the responsible mediator of anti-CTLA-4 therapy through inducting a Th1 immune response [19]. Interestingly, increase in the abundance of Bacteroides thetaiotaomicron was also found in EcN-treated mice compared to tumor bearing controls. In addition, Bifidobacterium was reported to facilitate the antitumor immune response to anti-PD-L1 therapy in melanoma bearing mice. [37]. However, the abundance of Bifidobacterium was not changed after EcN treatment in our study, suggesting that the antitumor effect of EcN did not follow the Bifidobacterium way and different bacteria may be associated with a better response to different immunotherapy. This discrepancy could be partly explained by different tumors with high heterogeneity, the different sources and genetic background of the mice enrolled, or the inter-population variability of the microbiota composition among them. Akkermansia muciniphila, as a conserved intestinal symbiont, has increasingly been studied and recognized as the gatekeeper of our mucosa promoting beneficial interactions in the intestinal tract [18]. Results found that the decreased relative abundance of Akkermansia muciniphila was correlated to liver diseases, inflammatory bowel diseases and many other diseases [41]. Furthermore, it is noteworthy that Akkermansia muciniphila has recently been associated with the clinical response to anti-PD1 immunotherapy [18]. Importantly, EcN-treated mice, in our study, displayed a higher relative abundance of Akkermansia muciniphila than tumor bearing controls, indicating that EcN therapy favors the dominance of selected Akkermansia species. The results discussed above undoubtedly place the gut microbiota in a synergic role in driving immunotherapy efficacy. Hence, strategies aimed at manipulating gut microflora are increasingly recognized as a useful strategy to improve therapeutic outcome.
Cancer immunotherapy has shown potential role in achieving long-lasting responses in a proportion of patients who fail in conventional therapies [2]. However, the efficacy is still limited, owing to the heterogeneity of the immune response among individuals and the distinct susceptibility of TME [6, 10, 11]. The cognition of gut microbiota in modulating immunotherapy response offers a new possibility to improve its therapeutic efficacy by targeting the microbiota [42]. Our work and other related studies have reported the important role of the certain specific bacteria in restore and sensitizing of tumor immunotherapy [14, 34, 43, 44]. For example, Lactobacillus rhamnosus GG is well characterized and has been shown to be helpful in anti-cancer therapeutic practice [44]. In particular, it can stimulate the DCs maturation and modulate T-cells mediated anti-tumor immune response [45]. However, the exact molecular mechanisms underlying certain specific species need further exploration. As for EcN, as one of the most investigated probiotic bacteria, numerous studies have demonstrated its beneficial effects in the management of intestinal inflammatory disorders and other probiotic properties. Nevertheless, the underlying mechanism is poorly understood and preclinical studies addressing its efficacy in cancer immunotherapy are still lacking. To this end, there is a need for further research and exploration of EcN and other bacteria in cancer treatment. In particular, more attention should be paid to the inter-individual variability in microbiota composition between animal models and clinical settings. To realize personalized strategy for microbiota optimization in precision medicine, microbial profiles of different individuals should be further explored. Herein, devoting more efforts in better elucidating the complex interactions between host and gut microbiota would pave a new path for improving cancer immunotherapy outcome.
Conclusion
In conclusion, our research unraveled that a specific commensal strain EcN was able to enhance the therapeutic efficacy of TGF-β blockade. Combination treatment with TGF-β blockade and EcN could markedly trigger tumor suppression and necrosis in subcutaneous tumor bearing mice, making it superior to treatment with TGF-β blockade or EcN alone. Meanwhile, we found that the enhanced therapeutic efficiency was accompanied by eliciting of tumor-specific effector T cells response and increased accumulation of antigen-specific IFN-γ+CD8+ T cells within the tumor. Notably, the antitumor function unfolded by EcN was likely to be mediated by the intestinal microbiota which played a synergic role in driving cancer immunotherapy efficacy. Hence, our findings provide a more comprehensive perspective to improve cancer immunotherapy outcome through manipulating the gut microflora, which may have important clinical implications for cancer prevention or treatment.
Abbreviations
ICIs: immune checkpoint inhibitors; rRNA: ribosomal RNA; EcN: Escherichia coli strain Nissle 1917; TGF-β: transforming growth factor-β; IFN-γ: interferon gamma; TME: tumor microenvironment; CTLs: cytotoxic T cells; Tregs: regulatory T cells; MDSC: myeloid-derived suppressor cells; TILs: tumor-infiltrating lymphocytes; MHC: major histocompatibility complex; DCs: dendritic cells; Th1: Type 1 T helper; Th17: T helper cell 17; NK: natural killer; ELISA: enzyme-linked immune sorbent assay; PLS-DA: partial least squares discriminant analysis; OTU: operational taxonomic unit; H&E: hematoxylin and eosin staining.
Supplementary Material
Supplementary figures.
Acknowledgements
This work was supported by grants from National Basic Research Program of China (2015CB931802), National Natural Science Foundation of China (81627901, 81703443, 81572050, 31770132 and 81873969). We thank Dr. Ying Liu and Shuyan Liang from Wuhan institute of biotechnology, Public Technology Service Platform for their kind help in flow cytometric analysis for experimental samples. We greatly appreciate the kind help of Dr. Zeneng Wang from Cleveland Clinic for revising the manuscript. We thank Xiaodong Jiang for his great help in animal experiments. We also thank the technical support of Dr. Min Lei from the Core Facilities for Life Science Research, Huazhong University of Science and Technology.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Gao S, Yang D, Fang Y, Lin X, Jin X, Wang Q. et al. Engineering Nanoparticles for Targeted Remodeling of the Tumor Microenvironment to Improve Cancer Immunotherapy. Theranostics. 2019;9:126-51
2. Littman DR. Releasing the Brakes on Cancer Immunotherapy. Cell. 2015;162:1186-90
3. Lizee G, Overwijk WW, Radvanyi L, Gao J, Sharma P, Hwu P. Harnessing the power of the immune system to target cancer. Annu Rev Med. 2013;64:71-90
4. Yang Y. Cancer immunotherapy: harnessing the immune system to battle cancer. J Clin Invest. 2015;125:3335-7
5. Topalian SL. Targeting Immune Checkpoints in Cancer Therapy. JAMA. 2017;318:1647-8
6. Weigmann K. Releasing the brakes to fight cancer: The recent discovery of checkpoints has boosted the field of cancer immunotherapy. EMBO Rep. 2016;17:1257-60
7. Neuzillet C, Tijeras-Raballand A, Cohen R, Cros J, Faivre S, Raymond E. et al. Targeting the TGFbeta pathway for cancer therapy. Pharmacol Ther. 2015;147:22-31
8. Bueno L, de Alwis DP, Pitou C, Yingling J, Lahn M, Glatt S. et al. Semi-mechanistic modelling of the tumour growth inhibitory effects of LY2157299, a new type I receptor TGF-beta kinase antagonist, in mice. Eur J Cancer. 2008;44:142-50
9. Rothenberg ML, Carbone DP, Johnson DH. Improving the evaluation of new cancer treatments: challenges and opportunities. Nat Rev Cancer. 2003;3:303-9
10. Yarchoan M, Hopkins A, Jaffee EM. Tumor Mutational Burden and Response Rate to PD-1 Inhibition. N Engl J Med. 2017;377:2500-1
11. Pitt JM, Vetizou M, Daillere R, Roberti MP, Yamazaki T, Routy B. et al. Resistance Mechanisms to Immune-Checkpoint Blockade in Cancer: Tumor-Intrinsic and -Extrinsic Factors. Immunity. 2016;44:1255-69
12. Akhurst RJ, Hata A. Targeting the TGFbeta signalling pathway in disease. Nat Rev Drug Discov. 2012;11:790-811
13. A study of galunisertib (LY2157299) in combination with nivolumab in advanced refractory solid tumors and in recurrent or refractory NSCLC, or hepatocellular carcinoma. 2015. https://clinicaltrials.gov/ct2/show/NCT02423343
14. Nelson MH, Diven MA, Huff LW, Paulos CM. Harnessing the Microbiome to Enhance Cancer Immunotherapy. J Immunol Res. 2015;2015:368736
15. Routy B, Gopalakrishnan V, Daillere R, Zitvogel L, Wargo JA, Kroemer G. The gut microbiota influences anticancer immunosurveillance and general health. Nat Rev Clin Oncol. 2018;15:382-96
16. Pope JL, Tomkovich S, Yang Y, Jobin C. Microbiota as a mediator of cancer progression and therapy. Transl Res. 2017;179:139-54
17. Schwabe RF, Jobin C. The microbiome and cancer. Nat Rev Cancer. 2013;13:800-12
18. Routy B, Le Chatelier E. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. 2018;359:91-7
19. Vetizou M, Pitt JM, Daillere R, Lepage P, Waldschmitt N, Flament C. et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science. 2015;350:1079-84
20. Sonnenborn U. Escherichia coli strain Nissle 1917-from bench to bedside and back: history of a special Escherichia coli strain with probiotic properties. FEMS Microbiol Lett. 2016:363
21. Secher T, Kassem S, Benamar M, Bernard I, Boury M, Barreau F. et al. Oral administration of the probiotic strain Escherichia coli nissle 1917 reduces susceptibility to neuroinflammation and repairs experimental autoimmune encephalomyelitis-induced intestinal barrier dysfunction. Front Immunol. 2017;8:1096
22. Scaldaferri F, Gerardi V, Mangiola F, Lopetuso LR, Pizzoferrato M, Petito V. et al. Role and mechanisms of action of Escherichia coli Nissle 1917 in the maintenance of remission in ulcerative colitis patients: An update. World J Gastroenterol. 2016;22:5505-11
23. Lasaro MA, Salinger N, Zhang J, Wang Y, Zhong Z, Goulian M. et al. F1C fimbriae play an important role in biofilm formation and intestinal colonization by the Escherichia coli commensal strain Nissle 1917. Appl Environ Microbiol. 2009;75:246-51
24. Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B. et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature. 2011;472:57-63
25. Dong W, Zhang H, Yin X, Liu Y, Chen D, Liang X. et al. Oral delivery of tumor microparticle vaccines activates NOD2 signaling pathway in ileac epithelium rendering potent antitumor T cell immunity. Oncoimmunology. 2017;6:e1282589
26. Maier A, Peille AL, Vuaroqueaux V, Lahn M. Anti-tumor activity of the TGF-beta receptor kinase inhibitor galunisertib (LY2157299 monohydrate) in patient-derived tumor xenografts. Cell Oncol (Dordr). 2015;38:131-44
27. Derynck R, Akhurst RJ, Balmain A. TGF-beta signaling in tumor suppression and cancer progression. Nat Genet. 2001;29:117-29
28. Herbertz S, Sawyer JS, Stauber AJ, Gueorguieva I, Driscoll KE, Estrem ST. et al. Clinical development of galunisertib (LY2157299 monohydrate), a small molecule inhibitor of transforming growth factor-beta signaling pathway. Drug Des Devel Ther. 2015;9:4479-99
29. Park J, Wrzesinski SH, Stern E, Look M, Criscione J, Ragheb R. et al. Combination delivery of TGF-beta inhibitor and IL-2 by nanoscale liposomal polymeric gels enhances tumour immunotherapy. Nat Mater. 2012;11:895-905
30. Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19:1423-37
31. McAllister SS, Weinberg RA. The tumour-induced systemic environment as a critical regulator of cancer progression and metastasis. Nat Cell Biol. 2014;16:717-27
32. de Visser KE, Eichten A, Coussens LM. Paradoxical roles of the immune system during cancer development. Nat Rev Cancer. 2006;6:24-37
33. Whiteside TL. The tumor microenvironment and its role in promoting tumor growth. Oncogene. 2008;27:5904-12
34. Vivarelli S, Salemi R, Candido S, Falzone L, Santagati M. Gut Microbiota and Cancer: From Pathogenesis to Therapy. Cancers (Basel). 2019:11 (1)
35. Tsilimigras MC, Fodor A, Jobin C. Carcinogenesis and therapeutics: the microbiota perspective. Nat Microbiol. 2017;2:17008
36. Viaud S, Saccheri F, Mignot G, Yamazaki T, Daillere R, Hannani D. et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science. 2013;342:971-6
37. Sivan A, Corrales L, Hubert N, Williams JB, Aquino-Michaels K, Earley ZM. et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. 2015;350:1084-9
38. Elkrief A, Derosa L, Zitvogel L, Kroemer G. The intimate relationship between gut microbiota and cancer immunotherapy. Gut Microbes. 2018:1-5
39. Hancock V, Dahl M, Klemm P. Probiotic Escherichia coli strain Nissle 1917 outcompetes intestinal pathogens during biofilm formation. J Med Microbiol. 2010;59:392-9
40. Iida N, Dzutsev A, Stewart CA, Smith L, Bouladoux N, Weingarten RA. et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science. 2013;342:967-70
41. de Vos WM. Microbe Profile: Akkermansia muciniphila: a conserved intestinal symbiont that acts as the gatekeeper of our mucosa. Microbiology. 2017;163:646-8
42. Chaput N, Lepage P, Coutzac C, Soularue E, Le Roux K, Monot C. et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann Oncol. 2017;28:1368-79
43. Lazar V, Ditu LM, Pircalabioru GG, Gheorghe I, Curutiu C, Holban AM. et al. Aspects of Gut Microbiota and Immune System Interactions in Infectious Diseases, Immunopathology, and Cancer. Front Immunol. 2018;9:1830
44. Banna GL, Torino F, Marletta F, Santagati M, Salemi R, Cannarozzo E. et al. Lactobacillus rhamnosus GG: An Overview to Explore the Rationale of Its Use in Cancer. Front Pharmacol. 2017;8:603
45. Cai S, Kandasamy M. Lactobacillus rhamnosus GG Activation of Dendritic Cells and Neutrophils Depends on the Dose and Time of Exposure. J Immunol Res. 2016;2016:7402760
Author contact
Corresponding authors: Email: bixiangzhangcom (B.X. Zhang), zhiliuedu.cn (Z. Liu), yangxlhust.edu.cn (X.L. Yang).
 Global reach, higher impact
Global reach, higher impact