Impact Factor
Theranostics 2019; 9(19):5672-5680. doi:10.7150/thno.33867 This issue Cite
Research Paper
Visualizing Autophagic Flux during Endothelial Injury with a Pathway-Inspired Tandem-Reaction Based Fluorogenic Probe
1. College of Pharmaceutical Sciences, Zhejiang University, Hangzhou 310058, China
2. State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai 201203, China
3. School of Pharmacy, Nanjing Medical University, Nanjing 211166, China
*These authors contributed equally
Abstract
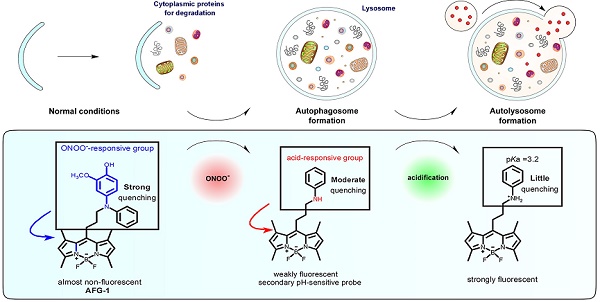
Autophagy is a dynamic and complicated catabolic process. Imaging autophagic flux can clearly advance knowledge of its pathophysiology significance. While the most common way autophagy is imaged relies on fluorescent protein-based probes, this method requires substantial genetic manipulation that severely restricts the application. Small fluorescent probes capable of tracking autophagic flux with good spatiotemporal resolution are highly demanable.
Methods: In this study, we developed a small-molecule fluorogenic probe (AFG-1) that facilitates real-time imaging of autophagic flux in both intact cells and live mice. AFG-1 is inspired by the cascading nitrosative and acidic microenvironments evolving during autophagy. It operates over two sequential steps. In the first step, AFG-1 responds to the up-regulated peroxynitrite at the initiation of autophagy by its diphenylamino group being oxidatively dearylated to yield a daughter probe. In the second step, the daughter probe responds to the acidic autolysosomes at the late stage of autophagy by being protonated.
Results: This pathway-dependent mechanism has been confirmed first by sequentially sensing ONOO- and acid in aqueous solution, and then by imaging autophagic flux in live cells. Furthermore, AFG-1 has been successfully applied to visualize autophagic flux in real-time in live mice following brain ischemic injury, justifying its robustness.
Conclusion: Due to the specificity, easy operation, and the dynamic information yielded, AFG-1 should serve as a potential tool to explore the roles of autophagy under various pathological settings.
Keywords: Fluorescent imaging, fluorescent probe, autophagy, endothelial injury
 Global reach, higher impact
Global reach, higher impact